Over the course of schizophrenia, there is typically a decrease in the size of brain structures. Intriguingly in terms of potential etiology, there is evidence of increased levels of mediators of inflammation and oxidative stress in the serum and plasma of affected individuals, when compared to healthy controls.
One of the first meta-analyses (by Potvin and colleagues in 2008) reported that of 19 studies investigating levels of interleukin-6 (IL-6) in schizophrenia, 16 had found at least a small increase relative to controls.1 Overall, Potvin et al also found increased levels of IL-1RA and sIL-2R, and a lower level of IL-2. They comment, however, that a role for disease-associated stress and weight gain in the abnormal cytokine profile of patients cannot be excluded. Subsequently, Miller et al reported higher levels of cytokines during an acute exacerbation of schizophrenia and noted that levels fall with treatment.2 There are similar findings with mediators of oxidative stress.3
Blood-borne factors related to inflammation and aging
Such findings have led authors to consider immune system-mediated mechanisms that may cause or contribute to psychosis.4
“Immune system-mediated mechanisms may cause or contribute to psychosis. Central to this is the activation of microglia.”
Central to this picture is the activation of microglia, leading to increased levels of the pro-inflammatory cytokines IL-6 and IL-1beta and Tumor Necrosis Factor-alpha, and a decrease in levels of the anti-inflammatory cytokine IL-10. This would result in turn in higher levels of oxidative stress and cortisol, the elaboration of auto-antibodies by B cells, and altered synaptic pruning.
The consequences would be abnormal neurodevelopment, neurodegeneration, and the presence of positive, negative and cognitive symptoms characteristic of schizophrenia.
Elements of this picture are supported by animal data, as in the work of Ribeiro et al (2013)5 who demonstrated progressive microglial activation and an increase in iNOS expression in rats subjected to a neurodevelopmental model of schizophrenia. Importantly, these changes could be reversed by clozapine.
Interest in blood-borne factors that may influence aging in people with schizophrenia was stimulated by the striking findings of Villeda et al who performed a series of experiments in which young mice shared the circulation of old animals.6 Young animals exposed to plasma from old mice showed age-related brain changes, notably decreased hippocampal neurogenesis and synaptic plasticity, and impaired memory.
The authors identified certain chemokines – including eotaxin (also known as CCL11) – whose plasma levels correlated with reduced neurogenesis in mice exposed to the blood of older animals. They also showed that increasing levels of circulating eotaxin in young mice resulted in decreased neurogenesis and cognitive deficits. Relevant to normal aging – and perhaps accelerated aging in schizophrenia – Villeda et al note that eotaxin levels in human plasma and CSF rise with age.
Pedrini et al assayed levels of eotaxin in the serum of people early or late in the course of schizophrenia, comparing each group to healthy controls.7 Early in the disease course, eotaxin levels were only marginally elevated relative to controls (Figure 1). However, the difference versus controls was large and significant in late-stage patients (who, typically, had had schizophrenia for twenty years).
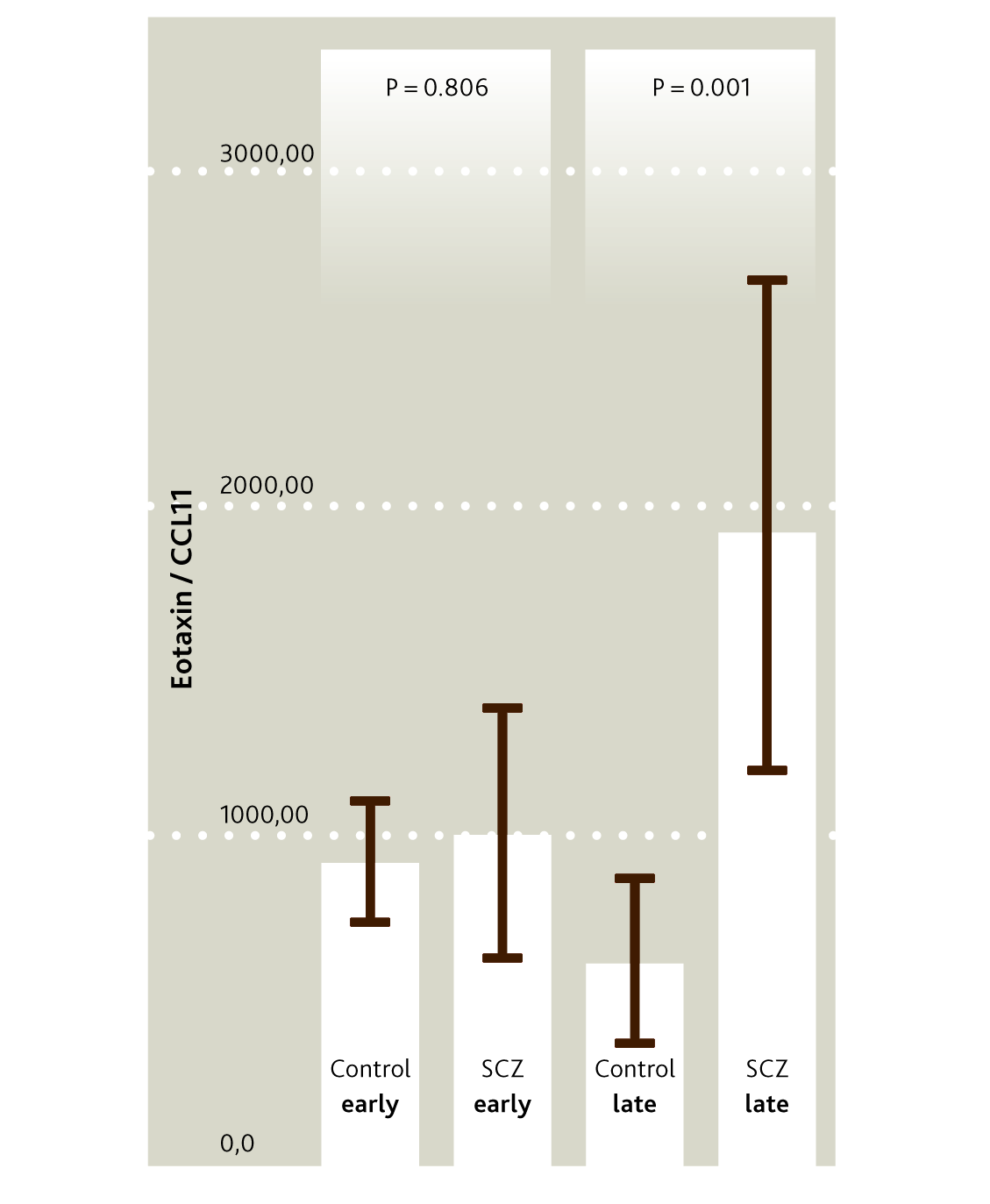
Figure 1
Serum eotaxin levels in patients with schizophrenia of recent onset and in patients with a history of chronic disease, versus controls.
In addition to its association with decreased hippocampal neurogenesis and impaired learning and memory in mice, eotaxin has chemo-attractant properties (for eosinophils, for example) relevant to inflammatory mechanisms.8
A tale told by telomeres
Since telomere erosion occurs with cell division, shortened telomere length is another characteristic of aging, though it can also be induced by environmental insults.9,10 In a meta-analysis of studies investigating leukocyte telomere length (LTL), Rao et al found that LTL among people with schizophrenia was significantly shorter than in controls – though the authors caution that all data were cross-sectional, and say that longitudinal studies are required to demonstrate progressive LTL erosion with longer disease course.11
Czepielewski et al compared healthy controls against people with schizophrenia (who had had the disorder for a mean of 14 years) and the siblings of those affected.12 Mean telomere length in people with schizophrenia was significantly shorter than in healthy controls but not shorter than in their siblings. The authors suggested that increased immune cell replication during inflammation might be the factor responsible for telomere shortening.
“Telomeres in people with schizophrenia were significantly shorter than in healthy controls but not shorter than in their siblings”
Heterogeneity within schizophrenia
Černis et al proposed that schizophrenia patients with high intelligence might form a distinct sub-type within the disorder.13 This prompted Gama and colleagues to investigate the possible relationship between intellectual functioning and cognitive performance and brain structure.
Brain imaging and related data were obtained from 94 patients included in the multi-site, open-access SchizConnect databank and compared with 94 healthy controls. Brain variables of interests were total and intracranial volume, gray matter and cortical thickness. Investigators assessed IQ using the vocabulary test of the Wechsler Adult Intelligence Scale III and cognition through measures of logical memory, letter-number sequencing and digit span.
On the basis of these scores, people with schizophrenia were divided into three groups: those with good IQ and good cognition, those with good IQ but poor cognition; and those with poor IQ and poor cognition. No patients had the combination of poor IQ but good cognition.
Using age and sex as covariates, patients with good IQ did not differ significantly from healthy controls in total intracranial volume (ICV). This was true irrespective of cognitive status. However, patients with both poor IQ and poor cognition had reduced ICV relative to other groups. Using age, sex and total brain volume as covariates, patients with good IQ and good cognition were again not different from healthy controls in total cortical gray matter volume and in cortical thickness. However, those with poor cognition – irrespective of IQ – had less gray matter than controls.
The same group has reported on 94 healthy controls and 92 people included in the Northwestern University Schizophrenia Data and Software Tool (NUSDAST).14 Subjects underwent neuropsychological assessment and structural MRI. Data from people with schizophrenia were analyzed by group according to cognitive performance and estimated premorbid IQ.
Lower IQ plus cognitive impairment was associated with smaller intracranial and total brain volume, smaller cortical gray matter volume, reduced cortical thickness and less insula volume. Subjects with poorer cognition but non-impaired IQ had only smaller total and cortical gray matter volume and reduced cortical thickness.
These different patterns of structural change led the authors to suggest that people who show impairment of both IQ and cognition have broad alterations in the brain indicative of early neurodevelopmental disruption. However, impaired cognition with intact premorbid IQ suggests cortical changes due primarily to neurodegeneration.
To see if the hypothesis of accelerated aging due to inflammation in people with schizophrenia would be supported, Gama and colleagues asked whether telomere length (a marker of aging), eotaxin (a marker of inflammation) and gray matter volume were more strongly related in people with the condition than in healthy controls.15 They looked at eotaxin levels in blood and structural MRI in 64 individuals with schizophrenia and 48 healthy controls.
“People with schizophrenia had decreased telomere length and gray matter, higher levels of eotaxin, and worse memory performance. These variables were not significantly correlated in healthy controls”
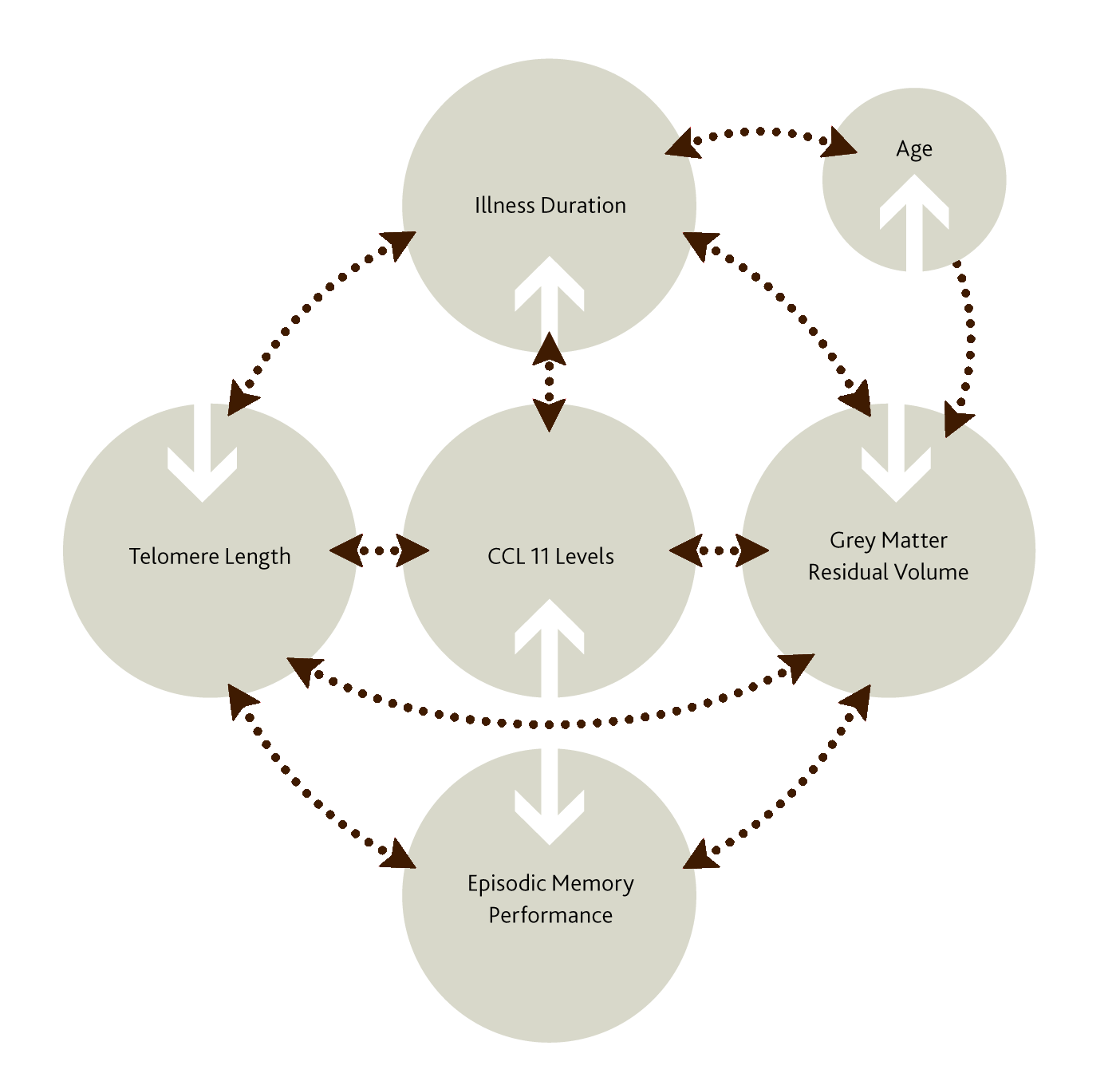
In people with schizophrenia, eotaxin levels were negatively correlated with telomere length (r= -.37, p=0.03). There was no significant correlation in controls. Similarly, in patients – but not in controls – gray matter volume correlated negatively with eotaxin levels. And again in patients – though not controls – gray matter volume correlated (this time positively) with telomere length. In patients, age was negatively correlated with telomere length, and illness duration with levels of eotaxin.
In patients with schizophrenia, we therefore seem to have a cluster of connections (see Figure 2) between longer duration of illness, raised levels of eotaxin, decreased telomere length, and reduced gray matter volume – and indeed also with impaired performance in episodic memory, which Gama and colleagues also investigated.
Summary
In conclusion, there is a pattern of results consistent with the idea that people with schizophrenia experience accelerated aging. This is reflected in reduced telomere length and possibly linked to chronic inflammation, and contributes to poor outcome including premature cognitive decline.
If these ideas can be substantiated by further work involving larger groups of patients and longitudinal studies, it would seem that Emil Kraepelin may have had a point when he popularized the term dementia praecox for the condition that became known as schizophrenia.
Access a collection of articles on Inflammation and Brain Disorders
Acknowledgments
We would like to thank Clarissa Gama, Universidad Federal do Rio Grande do Sul, Porto Alegre, Brazil, for sharing her expertise around accelerated aging in schizophrenia with us and for her support in developing this article.