Treatment of Schizophrenia and Dopamine Receptor Primacy
Although many different biological pathways are involved in schizophrenia and although multiple mechanisms of pharmacologic interventions for people with schizophrenia have been investigated, all currently approved medications for schizophrenia are antagonists or partial agonists at postsynaptic dopamine D2 receptors.1 However, the fact that still too many patients living with schizophrenia have residual and often functionally impairing positive, negative and cognitive symptoms calls for agents that have different mechanism of action (MOA) that can either be used in monotherapy or as augmentation of current dopamine receptor blockers.
The lack of novel MOA agents for the treatment of schizophrenia has not been due to lack of trying. Recently, the number of placebo-controlled randomized trials targeting postsynaptic dopamine receptors has been surpassed by agents with novel and different MOAs.2 A recent systematic review of phase-2 and phase-3 trials in adults with schizophrenia summarized agents targeting directly or indirectly diverse MOAs, including cannabinoid, cholinergic, dopamine, estrogen, GABA, glutamatergic, histamine, inflammatory, immunological, ion channel, melatonin, adrenaline, opioid, phosphodiesterase, serotonin, sigma, and trace amine associated receptor (TAAR) systems.3 Notably, however, of 176 completed or ongoing phase-2 or phase-3 trials, only 12 molecules, tested in 42 trials, outperformed placebo on the primary outcome.3
The lack of novel MOA agents for the treatment of schizophrenia has not been due to lack of trying.
Treatment of Schizophrenia Beyond Dopamine Receptors: Approved Agents
Despite multiple failed attempts at finding approvable treatments for schizophrenia with novel MOAs, there have been recently approved agents for schizophrenia by the US Food and Drug Administration (FDA) that go beyond dopamine receptor blockade. These new treatments include sublingual dexmedetomidine, a presynaptic alpha-2 receptor agonist reducing noradrenalin release, for the treatment of agitation in schizophrenia (and bipolar disorder),4 olanzapine-samidorphan combination that modulates opioid signaling reducing olanzapine’s associated weight gain retaining its efficacy in patients with schizophrenia (and bipolar disorder),5 and the vesicular monoamine transporter-2 (VMAT-2) inhibitors deutetrabenazine and valbenazine that reduce presynaptic dopamine availability for the treatment of tardive dyskinesia.6 The fact that VMAT-2 inhibitors reduce presynaptic dopamine, which has been related to the pathophysiology of schizophrenia more than postsynaptic dopamine dysfunction,1 coupled with antipsychotic effects of the irreversible VMAT-2 inhibitor, reserpine, suggest the need to test reversible VMAT-2 inhibition as a treatment for the acute and/or maintenance treatment of schizophrenia.7
Despite multiple failed attempts at finding approvable treatments for schizophrenia with novel MOAs, there have been recently approved agents for schizophrenia by the FDA that go beyond dopamine receptor blockade.
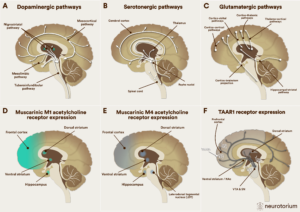
Figure 1: Neurotransmitter pathways involved in current and novel mechanism-action schizophrenia treatments
LDT=Laterodorsal tegmental nucleus; NAc = Nucleus Accumbens; SN = Substantia Nigra; VTA = Ventral Tegmental Area
A. Dopaminergic pathways are involved in positive, negative, and cognitive symptoms of schizophrenia and are affected by currently available first- and second-generation antipsychotics that act as dopamine antagonists or partial agonists and target psychosis.
B. Serotonergic pathways are involved in mood and psychosis and are affected by currently available second-generation antipsychotics that act as antagonists and/or partial agonists at several different serotonin receptors and target psychosis and mood symptoms.
C. Glutamatergic pathways are involved in positive, negative, and cognitive symptoms of schizophrenia and are affected by the M1/M4 receptor agonist xanomeline+tropsium targeting psychosis, and the glycine transporter-1 inhibitor, iclepertin, and the d-amino acid oxidase (DAOO) inhibitor luvadaxistat, each targeting cognition.
D. Muscarinic M1 acetylcholine pathways are involved in positive and cognitive symptoms of schizophrenia and are affected by the M1/M4 receptor agonist xanomeline+tropsium targeting psychosis and cognition.
E. Muscarinic M4 acetylcholine pathways are involved in positive symptoms of schizophrenia and are affected by the M1/M4 receptor agonist xanomeline+tropsium and by the M4 positive allosteric modulator (PAM) emraclidine, each targeting psychosis
F. The trace amine-associated receptor 1 (TAAR-1) system is mainly located intracellularly and interacts with trace amines, also called ”false” neurotransmitters, as they are not released from presynaptic terminals to stimulate postsynaptic receptors; instead when stimulated by trace amines or pharmacologic agonists, TAAR-1 receptors form heterodimer complexes with presynaptic and postsynaptic dopamine receptors, enhancing presynaptic dopamine autoreceptor activity, thereby reducing presynaptic dopamine synthesis, and reducing postsynaptic dopamine receptor availability and affinity for dopamine, reducing psychosis.
Read more about Novel and Emerging Pharmacologic Treatments for Schizophrenia
Treatment of Schizophrenia Beyond Dopamine Receptors: Agents in Late-Stage Clinical Development
Figure 1 displays the main neurotransmitter pathways involved in schizophrenia treatments currently available and of agents in late-stage clinical development. As recently reviewed,1 evidence for novel MOA agents, defined by ≥1 positive placebo-controlled trial, exists for the treatment of total symptoms (measured by the Positive and Negative Syndrome Scale [PANSS] total score) for the cannabinoid receptor agonist cannabidiol, D3 antagonist/5-HT1A partial agonist F17464, TAAR1and 5HT 1A agonist ulotaront, muscarinic M1/M4 agonist, xanomeline+trospium, and the M4 muscarinic positive allosteric modulator (PAM) emraclidine.3
Novel-MOA agents with preliminary evidence targeting negative symptoms include the 5-HT2A/sigma-2 antagonist roluperidone, and the 5-HT2A inverse agonist/antagonist and 5-HT2C antagonist pimavanserin that is FDA-approved for Parkinson’s disease psychosis.3
Agents with preliminary evidence targeting cognitive dysfunction associated with schizophrenia include the glycine transporter-1 inhibitor Iclepertin, and the d-amino acid oxidase (DAAO) inhibitor luvadaxistat.3
Finally, novel-MOA agents targeting residual positive symptoms/treatment-resistant schizophrenia include pimavanserin, the DAAO inhibitor sodium benzoate, the voltage-gated sodium channel blocker evenamide that modulates repetitive glutamate firing but not basal glutamate signaling, and xanomeline+trospium that is studied augmenting currently available antipsychotics for residual positive symptoms.3
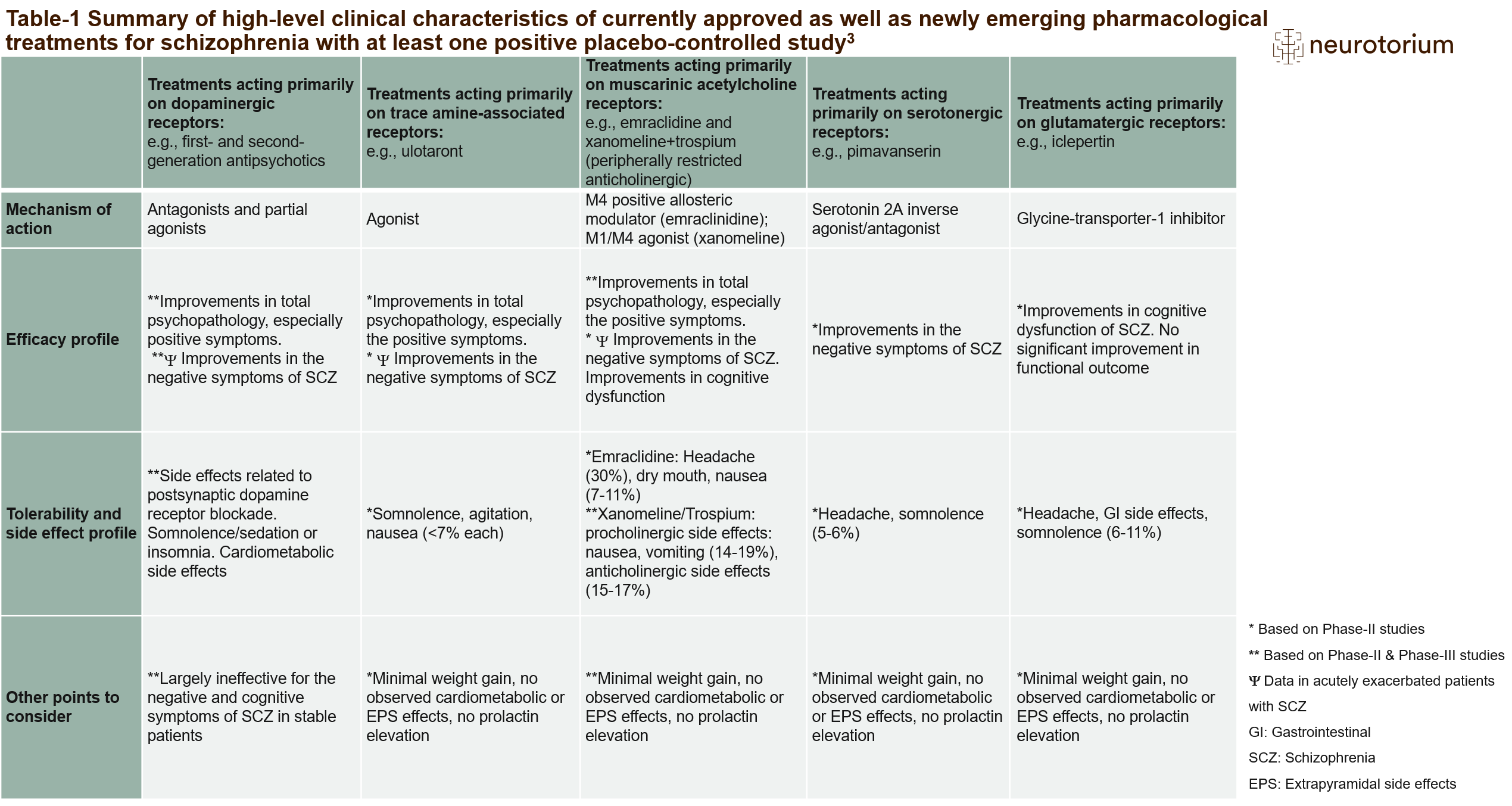
Table 1
The table summarizes the high-level clinical characteristics of the existing and newly emerging pharmacological treatments for schizophrenia with at least one positive placebo-controlled trial.
Improvements in positive symptoms of schizophrenia include hallucinations and delusions.
Improvements in negative symptoms of schizophrenia include affective flattening, alogia, anhedonia, motivation, and asociality.
Improvements in cognitive dysfunction include attention/vigilance, reasoning, problem-solving, processing speed, working memory, verbal learning and memory, visual learning and memory, and social cognition.
Extrapyramidal side effects (EPS) include dystonia, Parkinsonism, akathisia, and tardive dyskinesia.
Side effects related to postsynaptic dopamine receptor blockade include EPS, prolactin elevation, and sexual side effects.
Anticholinergic side effects include constipation and dyspepsia.
Cardiometabolic side effects include weight gain, dyslipidemia, and hyperglycemia.3
Table 1 summarizes high-level clinical characteristics of the currently approved dopamine antagonists/partial agonists as well as of several new MOA pharmacological treatments for schizophrenia with ≥1 positive placebo-controlled trial. Notably, none of the novel MOA agents directly block dopamine receptors. Therefore, none of these agents has relevant adverse effects related to dopamine receptor blockade, including neuromotor side effects, prolactin elevation, sedation, weight gain and glucose and lipid abnormalities.
Muscarinic receptor modulators: Clinical data
Among these novel MOAs explored for different symptom domains of schizophrenia, currently the most promising seems to be muscarinic system modulation. The xanomeline+trospium combination separated from placebo in three 5-week studies of patients with acutely exacerbated schizophrenia on the PANSS total score.8 Additional advantages in the acute trials were observed for negative symptoms, and for cognitive dysfunction in the subgroup of about 50% of patients with cognitive impairment at baseline. While improvements in negative symptoms and in cognitive dysfunction could still be “pseudo-specific”, i.e., be related to the concurrent improvements in other symptom domains, and while reasonably well-controlled placebo effects may also be responsible for the relatively higher effect sizes than observed in clinical trials of agents more recently approved for acute schizophrenia, data for muscarinic receptor enhancement as an effective MOA for the treatment of schizophrenia are robust. This includes both the efficacy of M1/M4 full agonism with xanomeline+trospium across 3 different clinical trials with consistent effect sizes for total PANSS symptoms of 0.60-0.758, and comparable efficacy with the M4 PAM, emraclidine, with effect sizes for total PANSS symptoms of 0.59-0.68 at week 6 in a small phase-1B study.10
However, since M1 receptors are also located in the periphery, xanomeline can lead to pro-cholinergic adverse effects, like nausea (17.5%) and vomiting (13.5%).8 To buffer these peripheral, undesired procholinergic effects, xanomeline was paired with the peripherally restricted anticholinergic trospium, which can lead to constipation (17.1%) and dyspepsia (15.3%). Nevertheless, these gastrointestinal adverse effects were mostly mild to moderate, seem to emerge mostly in the first few weeks, subsiding mostly within the first 3 weeks, and were rarely associated with treatment discontinuation.11 Being an M4 PAM, emraclidine has not been associated with peripheral pro-cholinergic effects and is not paired with a peripheral anticholinergic.
Among these novel MOAs explored for different symptom domains of schizophrenia, currently the most promising seems to be muscarinic system modulation.

Figure 2: M1 and M4 effects postulated to lead to reduced positive psychotic symptoms in schizophrenia
M4 receptors are presynaptic autoreceptors, and their stimulation inhibits acetylcholine release from the hindbrain to the ventral tegmental area in the midbrain. This reduced acetylcholine transmission reduces M5 receptor stimulation-related presynaptic dopamine synthesis in the associative striatum, where recent human evidence suggests that hyperdopaminergia is related to psychosis. Additionally, M4 autoreceptor agonism in the nucleus accumbens leads to reduced cholinergic interneuron stimulation of dopamine release and transmission in the associative striatum. Finally, M1 receptors are located postsynaptically in the frontal cortex and the hippocampus, among other brain areas. Stimulation of M1 receptors leads to the release of acetylcholine in the frontal cortex and the hippocampus, which can improve cognition. Stimulation of M1 receptors in layer II/III of the frontal cortex, however, increases acetylcholine, which in turn stimulates the inhibitory GABAergic interneuron. GABAergic stimulation then decreases glutamatergic transmission in layer V into the striatum, thereby reducing dopamine synthesis and transmission in the associative striatum.12,13
The highly encouraging data for xanomeline+trospium emraclidine suggest the emergence of at least one new medication class for the treatment of schizophrenia, i.e., muscarinic receptor enhancers. Different from current treatments for schizophrenia that are mostly receptor- and neurotransmitter-based, muscarinic receptor modulators are “system”-based treatments, in that they modulate dopamine synthesis and release with the modulation of acetylcholine, GABA and glutamate. Figure 2 summarizes the pharmacodynamic “bottom-up” system effects of M4 agonism/positive allosteric modulation as well as the “top-down” system effects of M1 agonism that are thought to be related to improvement in psychosis in patients with schizophrenia.12,13
Barriers to Progress
Despite the encouraging results of muscarinic receptor enhancers, barriers and questions regarding the successful development and implementation of novel MOA agents for the treatment of people with schizophrenia remain. High placebo effects are a major threat to successful trial results, especially when clinical trial development programs move from smaller, more controlled and positive phase-2 to larger phase-3 trials. Such a problem occurred recently with ulotaront, a TAAR-1/5HT1A agonist,14 where following a positive phase-2B study with an effect size versus placebo of 0.45, the two phase-3 ulotaront DIAMOND trials were negative. While the drug effect that was -17.2 points from baseline on the total PANSS scale in the phase-2B trial15 was largely replicated with between -16.4 and -19.6 PANSS points in the four ulotaront arms, the placebo improvement increased from -9.7 points in the phase-2 trial to -14.3 and -19.3 PANSS total points in the two phase-3 trials.16 This result raises the question whether prior novel MOA agents should be restudied if they had failed in the presence of high placebo response. Since the phase-2B study with ulotaront was conducted in patients aged ≤40 years and with ≤2 relapses lifetime, whereas the phase-3 trials enrolled patients aged ≤60 years and ≤3 prior relapses in one of the studies, this result raises the further question whether there could be biological subgroups in whom a specific novel MOA could be especially effective and whether novel MOA agents are more effective in patients with less prior dopamine receptor blockade or chronic illness effects.
Despite the encouraging results of muscarinic receptor enhancers, barriers and questions regarding the successful development and implementation of novel MOA agents for the treatment of people with schizophrenia remain.
Additional questions relate to desired rational polypharmacy options that are currently not available due to largely overlapping efficacy mechanisms of approved dopamine receptor blockers: could adding novel MOA agents to traditional antipsychotic medications be synergistic and lead to enhanced efficacy without undue adverse effect burden? Or, conversely, could postsynaptic and presynaptic dopamine blockade of currently available antipsychotics (with presynaptic dopamine blockade potentially disinhibiting presynaptic dopamine synthesis) mitigate or even abolish the efficacy of the novel MOA agent? Also, since all of the reviewed novel MOA agents tested for schizophrenia have mostly only short-term data available and in relatively small samples, could there be currently undetected, long-term, low-frequency risks? Furthermore, with different efficacy and side effect profiles, would possibly new rating scales be needed to better capture the effects of new MOA agents on the different domains of schizophrenia that they target? Moreover, could the advent of novel and distinct MOA agents prompt regulatory agencies to move from disease-based classifications and warnings to pharmacodynamic and MOA-based classifications, such as already described by the neuroscience-based nomenclature initiative?17 Finally, will there be global access to these novel MOA agents, given insurance constraints and added regulatory demands for example for studies with an active arm for assay sensitivity and demonstration of superiority in placebo-controlled relapse prevention studies before approval for an acute indication is granted by the European Medicine Agency?
Could the advent of novel and distinct MOA agents prompt regulatory agencies to move from disease-based classifications and warnings to pharmacodynamic and MOA-based classifications, such as already described by the neuroscience-based nomenclature initiative?
Summary
Although direct dopamine receptor modulation has been a crucial advancement in the management of schizophrenia, novel MOA agents that can effectively improve positive, negative and/or cognitive, as well as residual/refractory positive symptoms of schizophrenia are needed. Given that none of the reviewed studies of novel MOA agents included dopamine antagonist or partial agonist as control arm, the relative efficacy versus existing treatments needs to be established. Moreover, subgroup analyses should be performed to potentially identify clinical and/or biological characteristics that moderate efficacy of these novel MOA agents. Moving pharmacologically beyond dopamine receptor blockade is clearly important and much awaited. However, after approval of the first and hopefully of many novel MOA agents for the treatment of schizophrenia, including different symptom domains, next steps include demonstrating their real-world impact on outcomes beyond symptom control, like treatment adherence and satisfaction, functioning, quality of life and premature mortality.18-20
Although direct dopamine receptor modulation has been a crucial advancement in the management of schizophrenia, novel MOA agents that can effectively improve positive, negative and/or cognitive, as well as residual/refractory positive symptoms of schizophrenia are needed.
Declaration of Interest:
CU Correll has been a consultant and/or advisor to or has received honoraria from: AbbVie, Acadia, Alkermes, Allergan, Angelini, Aristo, Biogen, Boehringer-Ingelheim, Cardio Diagnostics, Cerevel, CNX Therapeutics, Compass Pathways, Darnitsa, Denovo, Gedeon Richter, Hikma, Holmusk, IntraCellular Therapies, Jamjoom Pharma, Janssen/J&J, Karuna, LB Pharma, Lundbeck, MedAvante-ProPhase, MedInCell, Merck, Mindpax, Mitsubishi Tanabe Pharma, Mylan, Neurocrine, Neurelis, Newron, Noven, Novo Nordisk, Otsuka, Pharmabrain, PPD Biotech, Recordati, Relmada, Reviva, Rovi, Sage, Seqirus, SK Life Science, Sumitomo Pharma America, Sunovion, Sun Pharma, Supernus, Takeda, Teva, Tolmar, Vertex, and Viatris. He provided expert testimony for Janssen and Otsuka. He served on a Data Safety Monitoring Board for Compass Pathways, Denovo, Lundbeck, Relmada, Reviva, Rovi, Supernus, and Teva. He has received grant support from Janssen and Takeda. He received royalties from UpToDate and is also a stock option holder of Cardio Diagnostics, Kuleon Biosciences, LB Pharma, Mindpax, and Quantic.