Metabolic Dysfunction in Schizophrenia
Schizophrenia is a chronic and severely debilitating psychiatric illness that affects approximately 1% of the population.1,2 Patients with schizophrenia are additionally afflicted by higher rates of cardiometabolic comorbidities compared to the general population, including nearly twice the rate of obesity, a 5-6-fold increased risk of developing type 2 diabetes mellitus, and a 3-6-fold increased risk of cardiovascular disease.3,4 Consequently, schizophrenia is associated with a 15-20-year shorter life expectancy than the general population.3,5 These cardiometabolic disturbances also have significant implications for self-esteem, quality of life, and cognitive functioning, worsening functional outcomes (Figure 1). Importantly, growing evidence highlights a close link between metabolic health and cognitive functioning; patients with comorbid metabolic syndrome perform worse on measures of cognition compared to those without metabolic syndrome.6-9 Furthermore, as illness course and antipsychotic treatment continue, the worsening metabolic profile has deleterious effects on cognitive functioning.10,11
Growing evidence highlights a close link between metabolic health and cognitive functioning.
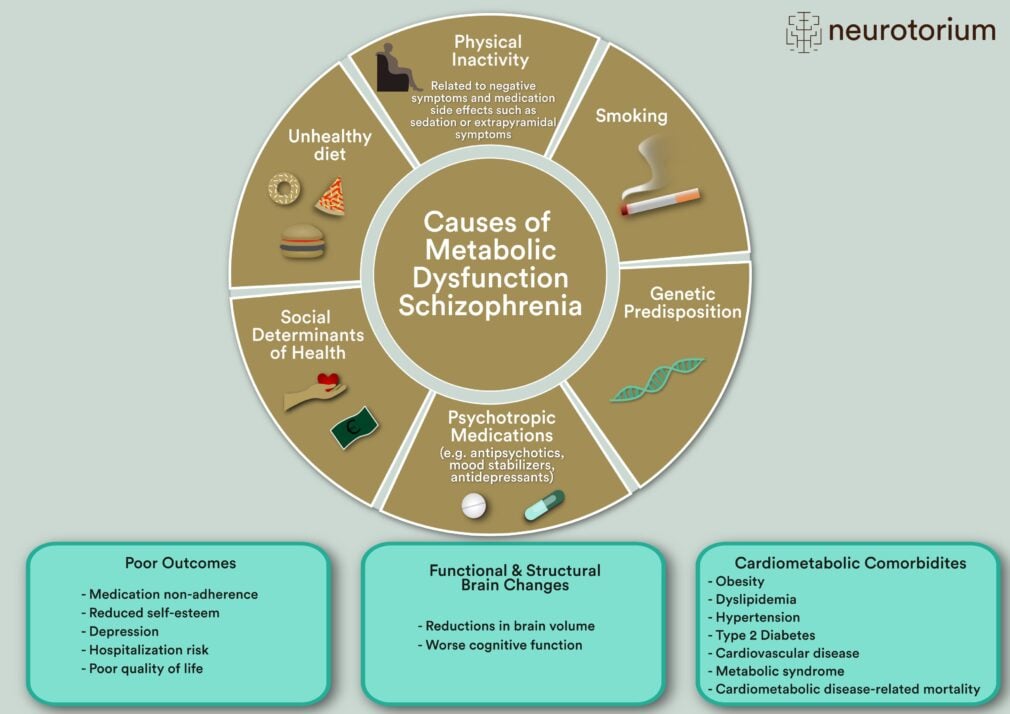
Figure 1. Causes and consequences of metabolic dysfunction in schizophrenia
Patients with schizophrenia experience high rates of cardiometabolic comorbidities such as obesity, dyslipidemia, hypertension, type 2 diabetes, and cardiovascular disease. The etiology of these comorbidities is multifactorial, arising from a complex interplay of intrinsic and extrinsic factors, including genetic predispositions, physical inactivity, unhealthy diet, high smoking rates, poorer socioeconomic status, as well as antipsychotic medication side effects. Beyond the devastating medical sequelae, these comorbidities also contribute to medication non-adherence, low self-esteem, decreased quality of life, and worse cognitive functioning.
The etiology of cardiometabolic comorbidities in schizophrenia is believed to be multifactorial, arising from a complex interplay of unhealthy lifestyle factors, reduced access to medical care, and metabolic side effects of antipsychotic medications. At the same time, the consistent presence of metabolic abnormalities in drug-naïve and first-episode patients suggests there may be an intrinsic vulnerability as well.12 This has led to the hypothesis that schizophrenia and metabolic disorders may share common pathophysiological mechanisms, and improving metabolic health may confer cognitive benefits.
This has led to the hypothesis that schizophrenia and metabolic disorders may share common pathophysiological mechanisms, and improving metabolic health may confer cognitive benefits.
Brain insulin action: a proposed unifying link underlying cognitive and metabolic dysfunction in schizophrenia
In recent years, brain insulin resistance has been positioned at the crossroads of cognitive and metabolic disorders.13 Brain insulin action is a novel area of investigation as the brain was long considered to be an insulin-insensitive organ. However, it is now well-recognized that insulin is also abundantly present within the central nervous system (CNS),14 with wide expression of insulin receptors in brain regions such as the hypothalamus, midbrain dopamine neurons, striatum, prefrontal cortex, and hippocampus.15 Insulin in the brain is involved in the regulation of several neuroendocrine, metabolic, and cognitive processes,13,16 many of which are notably disrupted in schizophrenia (Figure 2).
For example, insulin action within the hypothalamus suppresses hepatic glucose production,17-19 promotes satiety, reduces appetite, and increases energy expenditure.20,21 Insulin also modulates mood, supports memory formation through synaptic plasticity and stimulating long-term potentiation, and promotes neuronal survival by triggering the release of neurotrophic factors.16,22-25

Figure 2. Brain insulin action in relation to peripheral and central outcomes.
Insulin receptors are widely expressed throughout the brain, with high concentrations in regions such as the prefrontal cortex, striatum, hippocampus, hypothalamus and amygdala. Insulin acting within these brain regions exerts several downstream central and peripheral actions.
Causes of impaired brain insulin signaling in schizophrenia
Our understanding of impaired brain insulin action in schizophrenia is still in its infancy; however, it is likely a multilevel process involving both intrinsic illness-related mechanisms and antipsychotic exposure (Figure 3).23
Genetic/neurobiological causes of brain insulin resistance: Several genetic studies show overlap between schizophrenia and diabetes, with shared variants in genes regulating insulin signaling, including the Akt pathway, which is involved in glucose homeostasis. Reduced Akt1 activation, mRNA expression, and protein levels have been observed in the hippocampus, frontal cortex, and blood of patients with schizophrenia compared to healthy controls.23 In parallel, mitochondrial dysfunction, increased oxidative stress, and decreased levels of antioxidant products in schizophrenia may disrupt neuronal insulin sensitivity by damaging membrane lipids and insulin receptor components, as well as impairing intracellular signaling cascades such as the Akt pathway.26
Reduced Akt1 activation, mRNA expression, and protein levels have been observed in the hippocampus, frontal cortex, and blood of patients with schizophrenia compared to healthy controls.23
Peripheral causes of brain insulin resistance: In addition, several systemic metabolic disturbances are present in schizophrenia that act synergistically to drive central insulin resistance. For example, schizophrenia is characterized by greater inflammation and elevated inflammatory markers, including C-reactive protein (CRP), interleukin (IL)-6, and tumor necrosis factor-alpha (TNF-α).27 These have been implicated in the pathogenesis of insulin resistance and hyperinsulinemia. Hypothalamic-pituitary-adrenal (HPA)-axis dysregulation, characterized by abnormal cortisol release and stress response, further perpetuates inflammation and oxidative stress.28 Altogether, these disruptions contribute to the development of metabolic abnormalities, including peripheral insulin resistance that eventually leads to brain insulin resistance.23,29
Hypothalamic-pituitary-adrenal (HPA)-axis dysregulation, characterized by abnormal cortisol release and stress response, further perpetuates inflammation and oxidative stress.28

Figure 3. Central and peripheral causes of brain insulin resistance in schizophrenia
Multiple interconnected pathways may underlie impaired brain insulin signaling in schizophrenia. These include both central/neurobiological mechanisms (i.e., neuroinflammation, mitochondrial dysfunction, Akt pathway dysfunction, oxidative stress) and peripheral mechanisms (i.e., visceral adiposity, increased insulin release, chronic stress, and inflammation). Additionally, antipsychotic medications exacerbate both peripheral and central mechanisms, contributing to increased appetite and weight gain, increased insulin release, increased hepatic glucose production, and decreased glucose uptake by skeletal muscle.
Consequences of impaired brain insulin signaling in schizophrenia
Brain consequences: Insulin in the brain increases dopamine transporter (DAT) surface expression and dopamine reuptake in the ventral tegmental area and striatum.26,30,31 Impaired insulin action may contribute to increased dopamine availability, which in turn may relate to the development of positive symptoms.32 Central insulin dysregulation may also underlie the close relationship between cognitive dysfunction in mental illness. To this point, intranasal insulin (INI), a way of directly stimulating the brain with insulin, has been shown to improve verbal memory and visuospatial function, in healthy controls and in Alzheimer’s disease.33,34 Interestingly, despite demonstrated improvements with INI in these cognitive domains in other populations, no beneficial effects were observed in the three studies published to date exploring the use of INI in antipsychotic-treated patients with schizophrenia.35-37 These findings may allude to brain insulin resistance in this population and the postulated anti-insulin effects of antipsychotics in the brain, which may limit pro-cognitive effects of various interventions.38
Peripheral consequences: In line with the physiological effects of insulin action in the brain, central insulin resistance has been associated with long-term adiposity, glucose dysregulation, and reduced peripheral insulin sensitivity, thus implicating it in the development of metabolic disorders such as obesity and diabetes.39 As such, this phenotype may help explain the alarmingly high rates of metabolic disorders seen in people with schizophrenia. It is important to recognize the circular relationship between peripheral and central insulin resistance. Central insulin resistance impairs appetite control and glucose regulation, promoting weight gain and peripheral insulin resistance. At the same time, peripheral insulin resistance leads to hyperinsulinemia and inflammation which can compromise blood-brain barrier integrity and insulin signaling in the brain.
It is important to recognize the circular relationship between peripheral and central insulin resistance.
The contributing role of antipsychotics on impaired brain insulin signaling
Antipsychotic treatment may exacerbate intrinsic brain insulin resistance though direct effects on insulin signaling pathways, as well as through the secondary effects of peripheral metabolic abnormalities. For example, D2 antagonism by antipsychotics results in an increase hypothalamic AMPK activation and inhibits the ability of central insulin to suppress endogenous glucose production.40,41 At the same time, antipsychotics also act directly on peripheral D2-receptors expressed on pancreatic beta cells.42 These receptors act as autocrine regulators to inhibit glucose-stimulated insulin secretion. Consequently, antipsychotics may induce hyperinsulinemia, which has been demonstrated in healthy control studies with acute antipsychotic administration.43 This hyperinsulinemia may lead to insulin resistance, hinder insulin transport across the blood-brain barrier, and impair central insulin action and its downstream effects. Furthermore, preclinical studies have demonstrated that central infusion of olanzapine results in an acute upregulation of NPY and AgRP mRNA expression.40 Upregulation of NPY may subsequently lead to disruptions in central insulin action and its physiologic suppression of appetite and endogenous glucose production. Finally, while increased inflammation may be intrinsic to the illness itself, antipsychotics may also contribute to and exacerbate the inflammation seen in patients, which in turn may potentiate weight gain and insulin resistance.44 Overall, these illness-related and treatment-induced processes converge to create a state of impaired insulin action which may contribute to greater cognitive dysfunction and heightened metabolic risk in this population.
Increased inflammation may be intrinsic to the illness itself, antipsychotics may also contribute to and exacerbate the inflammation seen in patients, which in turn may potentiate weight gain and insulin resistance.44
Therapeutic Implications and Future Directions
Deficits in brain insulin signaling present a unique opportunity that can be leveraged for both mechanistic inquiry and the development of novel therapeutic strategies in schizophrenia.
From the research standpoint, exploring brain insulin signaling can offer valuable insights into the pathophysiology of schizophrenia and metabolic comorbidities. However, studying insulin action in the human brain is still quite new and is limited to the use of proxy indicators. Multi-modal neuroimaging, combined with INI as a pharmacological challenge, is currently the most widely-published way to reliably, non-invasively, and safely examine the effects of insulin in the brain across healthy controls and various patient populations (e.g., obesity, type 2 diabetes, and Alzheimer’s disease).45 Approaches include resting state functional magnetic resonance imaging (rs-fMRI), arterial spin labeling (ASL), and task-based fMRI. Interestingly, these MRI-based outcome measures have been correlated with physiological and behavioural changes as well. For example, INI has been found to increase functional connectivity between the anterior medial prefrontal cortex and hypothalamus, but only in individuals with preserved peripheral insulin sensitivity. INI has also consistently been found to reduce regional cerebral blood flow (rCBF) in the hypothalamus,46,47 and this change in hypothalamic blood flow has been positively correlated with change in insulin sensitivity.48 Thus, this neuroimaging-based assay of brain insulin action can be utilized to further our understanding of impaired brain insulin signaling in relation to metabolic outcomes in schizophrenia relative to healthy individuals, and to disentangle the potential modulatory effects of antipsychotic medications.
From a clinical standpoint, this paradigm can also be used to assess whether metabolic interventions may reverse or improve brain insulin resistance in patients with schizophrenia and subsequently result in cognitive and metabolic improvements. Central insulin-sensitizers (i.e., drugs that increase the availability of insulin in the brain) such as metformin and glucagon-like-peptide 1 receptor agonists (GLP-1RAs) stand out as potential candidates for this purpose. Indeed, these agents have been investigated in antipsychotic-treated patients with schizophrenia and have shown efficacy in reducing body weight and mitigating other metabolic aberrations.49,50 Future treatment studies may consider assessing whether these medications intended for cardiometabolic improvement have potential neuroprotective effects as well.
INI has been found to increase functional connectivity between the anterior medial prefrontal cortex and hypothalamus, but only in individuals with preserved peripheral insulin sensitivity.
Conclusions
To summarize, growing evidence suggests that brain insulin resistance may play a key role in the pathophysiology of schizophrenia, particularly in relation to cognitive dysfunction, dopaminergic dysregulation, and the high burden of metabolic comorbidity. This represents a promising area of investigation in schizophrenia research that can provide us with greater insights into the underlying illness neurobiology and initiate new streams of work to uncover novel therapeutic strategies to treat both the illness and its metabolic comorbidities.
Growing evidence suggests that brain insulin resistance may play a key role in the pathophysiology of schizophrenia, particularly in relation to cognitive dysfunction, dopaminergic dysregulation, and the high burden of metabolic comorbidity.







