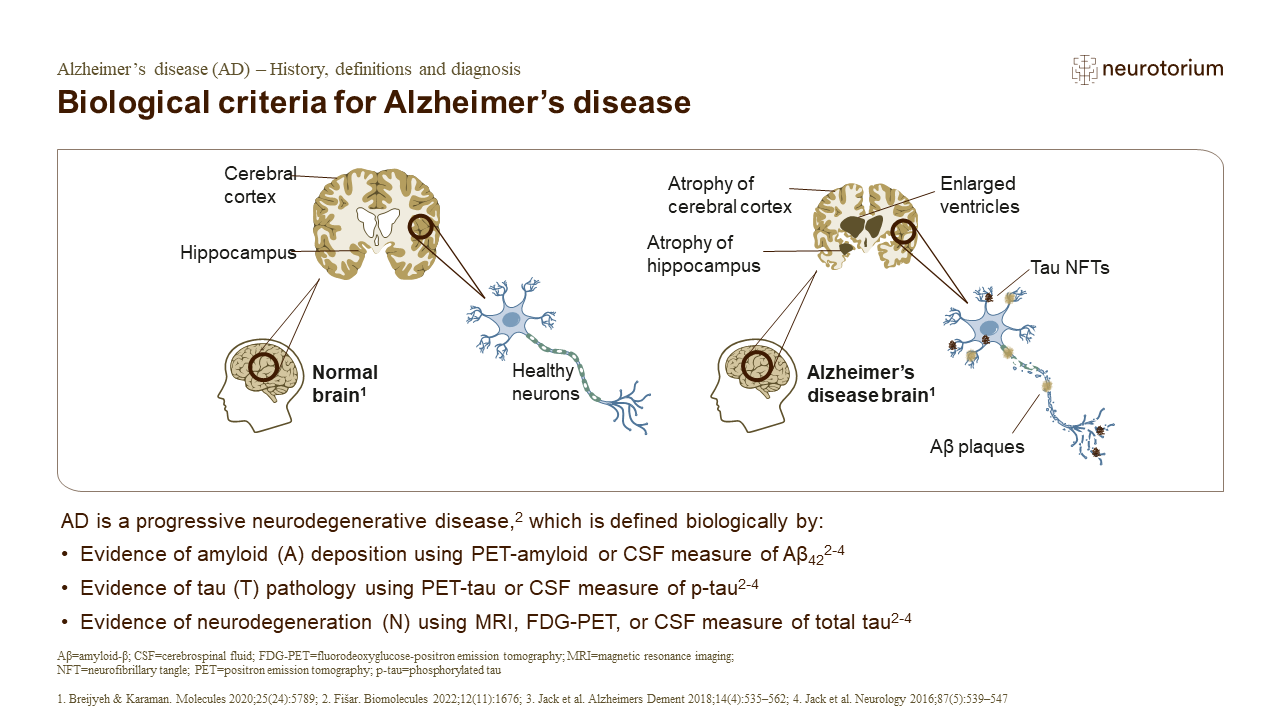
The temporal development of biomarker abnormalities can help understand the aetiology of AD and progression of disease.2 The A/T/N biomarker classification system for AD includes three main classes of biomarkers – Aβ, tau, and neurodegeneration – and provides the ability to classify biomarker status of an individual independently of their cognitive status.5 Ab biomarkers include amyloid positron emission tomography (PET) and cerebrospinal fluid (CSF) concentrations of Aβ.5 Tau biomarkers include tau-PET and CSF concentrations of phosphorylated p-tau.5 There are multiple neurodegeneration biomarkers, including fluorodeoxyglucose (FDG)-PET, magnetic resonance imaging (MRI) atrophy, CSF concentrations of total tau and of neurofilament light chain.5
In the preclinical phase, the first measurable evidence of AD is an abnormal reduction in Aβ1-42 levels within the CSF due to an increased sequestering of Ab in the brain parenchyma.6-9 Following this, Aβ tracer retention is observed in PET scans, directly reflecting the build-up of amyloid.9,10 As AD progresses, neurodegeneration and synaptic dysfunction can be observed using structural and functional neuroimaging techniques, such as FDG-PET and MRI.9,10 In carriers of the e4 APOE allele, synaptic dysfunction may be observed before detectable Aβ accumulation.10 Following this, elevated concentrations of CSF biomarkers that are directly associated with neuronal injury, such as t-tau, p-tau, visinin-like protein 1 (VILIP-1) and neurogranin, are detected.10,11 Continuation of disease results in hippocampal and entorhinal cortex atrophy and cortical thinning that can be detected by volumetric MRI.7,9,11 As with Ab biomarkers, these biomarkers may be detected in individuals at high risk of AD many decades before cognitive decline occurs.9,12 The MCI phase represents the first symptomatic occurrence in AD, with patients experiencing a decline or plateau in Aβ accumulation,12,15 and continued tau accumulation and neurodegeneration.9 The progression of AD pathology results in a display of cognitive deficits, with memory and executive function often being the most affected cognitive domains.9 For most patients with MCI due to AD pathology, the subsequent stage is AD dementia, which is a decline in clinical function, and represents the late stages of disease.9 At this stage, Aβ and tau accumulation are at their maximum, changes in brain structure continue, and neurodegeneration becomes macroscopically evident.9
References:
1.Breijyeh Z, Karaman R. Comprehensive review on Alzheimer’s disease: causes and treatment. Molecules 2020; 25 (24): 5789.
2.Fišar Z. Linking the amyloid, tau, and mitochondrial hypotheses of Alzheimer’s Disease and identifying promising drug targets. Biomolecules 2022; 12 (11): 1676.
3.Jack CR Jr, Bennett DA, Blennow K, et al. NIA-AA Research Framework: toward a biological definition of Alzheimer’s disease. Alzheimers Dement 2018; 14 (4): 535–562.
4.Jack CR Jr, Bennett DA, Blennow K, et al. A/T/N: An unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology 2016; 87 (5): 539–547.
5.Therriault J, Zimmer ER, Benedet AL, et al. Staging of Alzheimer’s disease: past, present, and future perspectives. Trends Mol Med 2022; 28 (9): 726–741.
6.Milà-Alomà M, Salvadó G, Gispert JD, et al; ALFA study. Amyloid beta, tau, synaptic, neurodegeneration, and glial biomarkers in the preclinical stage of the Alzheimer’s continuum. Alzheimers Dement 2020; 16 (10): 1358–1371.
7.Hadjichrysanthou C, Evans S, Bajaj S, et al; Alzheimer’s Disease Neuroimaging Initiative. The dynamics of biomarkers across the clinical spectrum of Alzheimer’s disease. Alzheimers Res Ther 2020; 12 (1): 74.
8.Palmqvist S, Mattsson N, Hansson O; Alzheimer’s Disease Neuroimaging Initiative. Cerebrospinal fluid analysis detects cerebral amyloid-β accumulation earlier than positron emission tomography. Brain 2016; 139 (Pt 4): 1226–1236.
9.Liss JL, Seleri Assunção S, Cummings J, et al. Practical recommendations for timely, accurate diagnosis of symptomatic Alzheimer’s disease (MCI and dementia) in primary care: a review and synthesis. J Intern Med 2021; 290 (2): 310–334.
10.Hampel H, Hardy J, Blennow K, et al. The amyloid-β pathway in Alzheimer’s Disease. Mol Psychiatry 2021; 26 (10): 5481–5503.
11.Jack CR Jr, Knopman DS, Jagust WJ, et al. Tracking pathophysiological processes in Alzheimer’s disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol 2013; 12 (2): 207–216.
12.Sperling R, Mormino E, Johnson K. The evolution of preclinical Alzheimer’s disease: implications for prevention trials. Neuron 2014; 84 (3): 608–622.
13.Fagan AM, Xiong C, Jasielec MS, et al. Longitudinal change in CSF biomarkers in autosomal–dominant Alzheimer’s disease. Sci Transl Med 2014; 6 (226): 226ra30.
14.Sperling RA, Aisen PS, Beckett LA, et al. Toward defining the preclinical stages of Alzheimer’s disease: recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimers Dement 2011; 7 (3): 280–292.
15.Jack CR Jr, Wiste HJ, Lesnick TG, et al. Brain β-amyloid load approaches a plateau. Neurology 2013; 80 (10): 890–896.