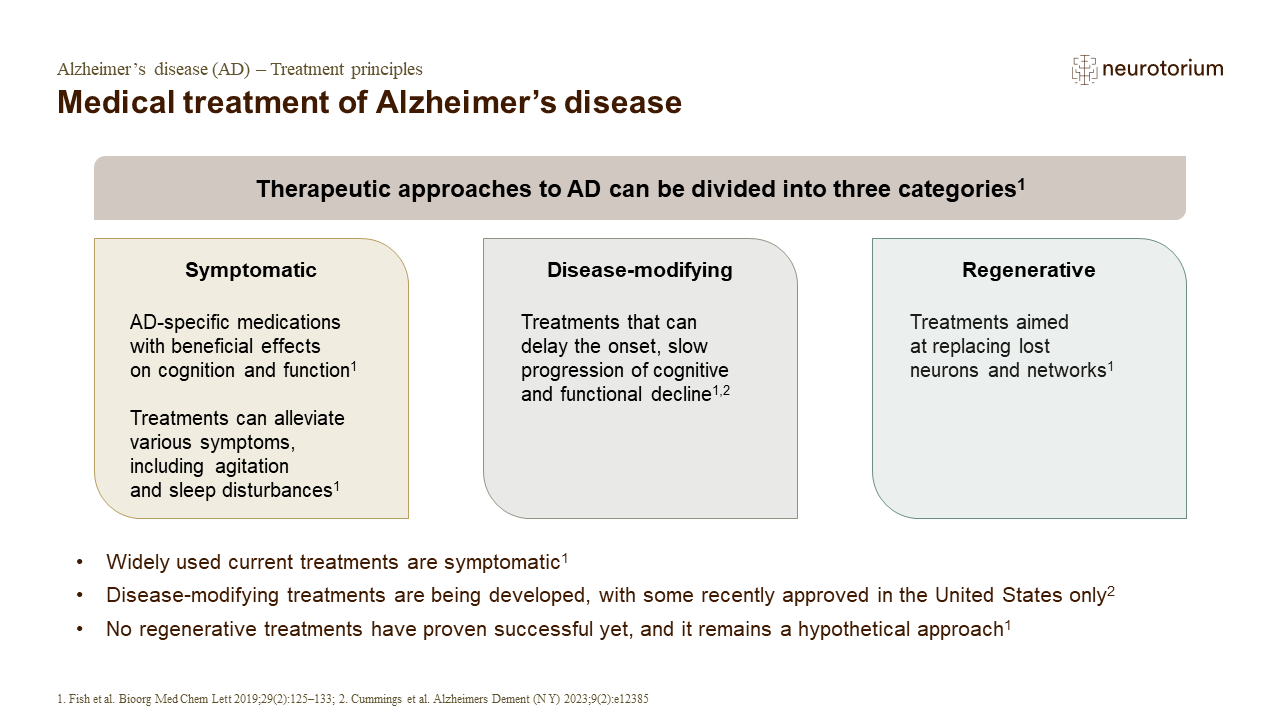
Therapeutic approaches to AD can be divided into three categories: symptomatic, disease-modifying and regenerative. In 2021, aducanumab received accelerated US Food and Drug Administration (FDA) approval for the treatment of AD.2 In 2023, lecanemab received accelerated FDA approval for the treatment of AD, which was later converted to traditional approval.2,3 However, in August 2023, these therapies were the only two drugs to have been approved since 2003.2-4
3. FDA press release 6 July 2023. FDA converts novel Alzheimer’s disease treatment to traditional approval. Available at: https://www.fda.gov/news-events/press-announcements/fda-converts-novel-alzheimers-disease-treatment-traditional-approval. Accessed 9 August 2023.
4. FDA press release 7 June 2021. FDA’s decision to approve new treatment for Alzheimer’s disease. Available at: https://www.fda.gov/drugs/news-events-human-drugs/fdas-decision-approve-new-treatment-alzheimers-disease. Accessed 9 August 2023.