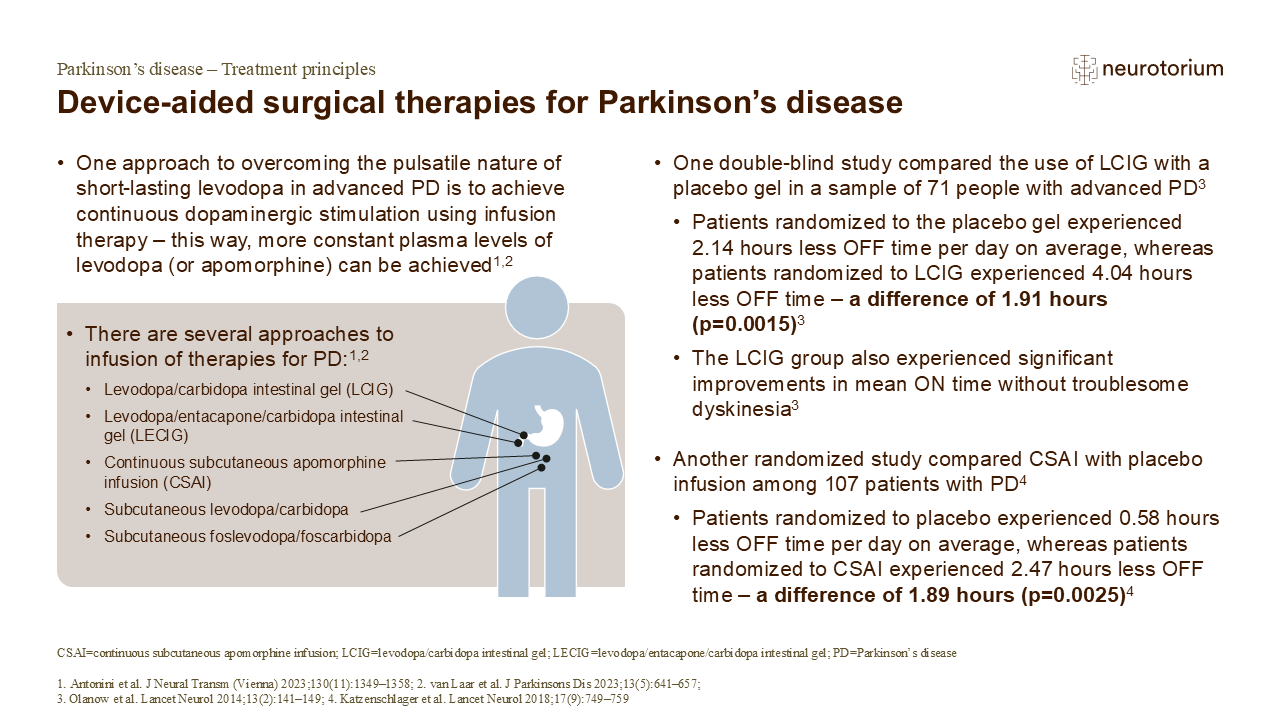
As outlined on the slide, device-aided therapy can allow a patient with Parkinson’s disease to achieve more stable plasma levels of levodopa or dopamine agonist than would be possible with oral therapy.1 Several different approaches to infusion therapy have been shown to be efficacious in the treatment of Parkinson’s disease, including levodopa/carbidopa intestinal gel (LCIG), continuous subcutaneous apomorphine infusion (CSAI), continuous subcutaneous levodopa-carbidopa infusion, and subcutaneous foslevodopa/foscarbidopa (a soluble formulation of levodopa and carbidopa prodrugs).3-6
References:
1. Antonini A, D’Onofrio V, Guerra A. Current and novel infusion therapies for patients with Parkinson’s disease. J Neural Transm (Vienna) 2023; 130 (11): 1349–1358.
2. van Laar T, Chaudhuri KR, Antonini A, et al. Infusion therapies in the treatment of Parkinson’s disease. J Parkinsons Dis 2023; 13 (5): 641–657.
3. Olanow CW, Kieburtz K, Odin P, et al. Continuous intrajejunal infusion of levodopa-carbidopa intestinal gel for patients with advanced Parkinson’s disease: a randomised, controlled, double-blind, double-dummy study. Lancet Neurol 2014; 13 (2): 141–149.
4. Katzenschlager R, Poewe W, Rascol O, et al. Apomorphine subcutaneous infusion in patients with Parkinson’s disease with persistent motor fluctuations (TOLEDO): a multicentre, double-blind, randomised, placebo-controlled trial. Lancet Neurol 2018; 17 (9): 749–759.
5. Espay AJ, Stocchi F, Pahwa R, et al. Safety and efficacy of continuous subcutaneous levodopa–carbidopa infusion (ND0612) for Parkinson’s disease with motor fluctuations (BouNDless): a phase 3, randomised, double-blind, double-dummy, multicentre trial. Lancet Neurol 2024; 23 (5): 465–476.
6. Soileau MJ, Aldred J, Budur K, et al. Safety and efficacy of continuous subcutaneous foslevodopa-foscarbidopa in patients with advanced Parkinson’s disease: a randomised, double-blind, active-controlled, Phase 3 trial. Lancet Neurol 2022; 21 (12): 1099–1109.