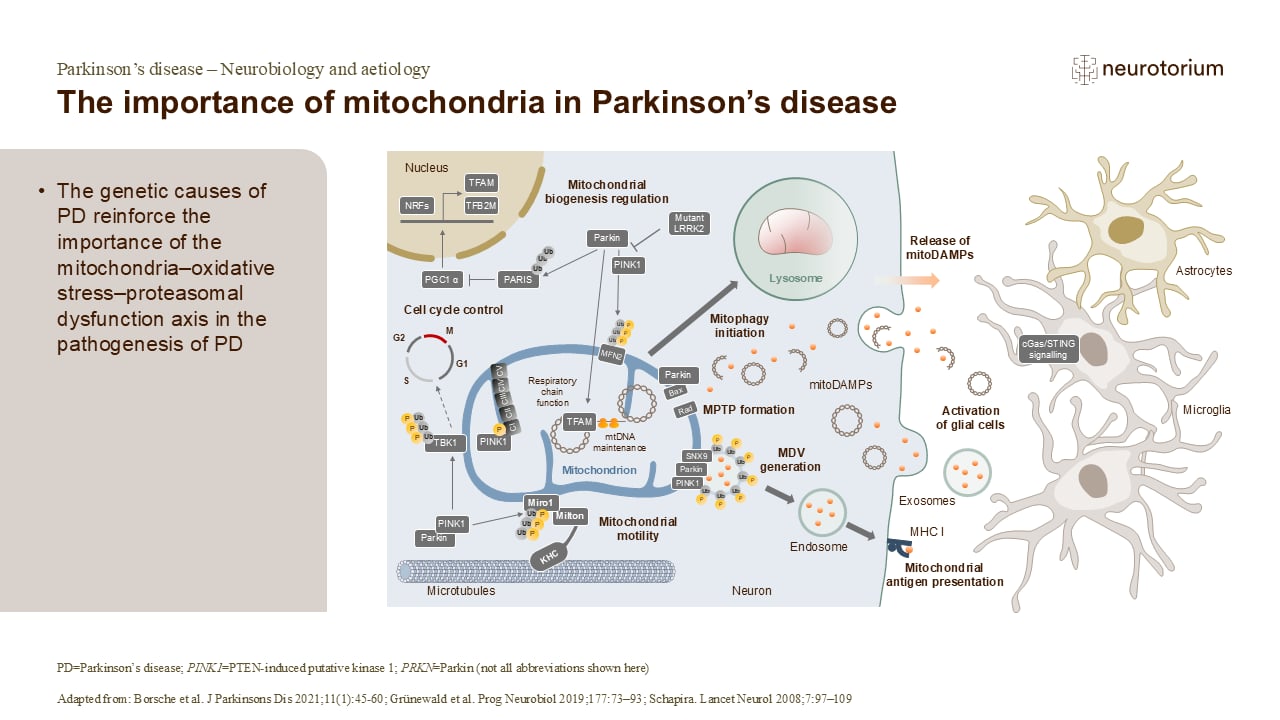
Mitochondria are known as the ‘power plants of the cell’. They use metabolic fuels such as glucose to produce the high-energy molecule, adenosine triphosphate (ATP), which drives many important physiological processes.
A toxic by-product of normal mitochondrial function is the generation of free radicals, which cause oxidative stress.4 The cellular responses to this oxidative stress are regulated by the genes DJ-1, PINK1, PRKN, and UCH-L1, all of which have genetic variations associated with PD.1-3 Taken together, the evidence strongly suggests that mitochondrial dysfunction plays an important role in the pathogenesis of PD.1,2 This opens the potential for novel treatments that may correct this dysfunction in people with PD.1