Reproductive aging
Reproductive aging, the gradual, physiological decline in ovarian function, is inevitable. Staging systems have provided a nomenclature and framework for reproductive ageing. The most widely used staging system is the Stages of Reproductive Aging Workshop + 10 (STRAW +10).1 Given the lack of standardisation in reproductive biomarkers, STRAW+10 relies primarily on the menstrual cycle characteristics (i.e., uterine bleeding) to define the different phases of reproductive aging. Its staging system is centred around the final menstrual period, the last time a period occurs before menopause, and spans from the reproductive years through the menopausal transition to postmenopause.
Hormones implicated in the physiology of reproductive aging include the Anti- Müllerian hormone (AMH), inhibin B, and the follicle-stimulating hormone (FSH). AMH is produced by small, growing egg follicles and is considered a marker of ovarian reserve, i.e., the number of egg follicles in the ovaries. Inhibin B is produced by larger, growing egg follicles in response to FSH secreted by the pituitary. In a negative feedback loop, inhibin B reduces FSH (Figure 1).
Changes in endocrine function and ovarian reserve begin before overt changes in menstrual cycles in the late reproductive stages.2 Hormonal changes in this phase compensate for the decline in ovarian reserve to maintain regular cycles. During the late reproductive stages, concentrations of AMH and inhibin B decrease, while FSH concentrations are either normal or inconsistently elevated.
The menopausal transition is characterised by markedly increased variability in menstrual cycle length and is usually divided into an early and late phase. In the early menopausal transition, defined by more than a 7-day difference in cycle length between consecutive menstrual cycles, levels of inhibin B and AMH continue to decrease. As inhibin B suppresses the secretion of FSH from the pituitary, its decline is associated with an increase in FSH. The duration of the early menopausal transition varies across people, with earlier onsets associated with a longer transition.1
Changes in endocrine function and ovarian reserve begin before overt changes in menstrual cycles in the late reproductive stages2
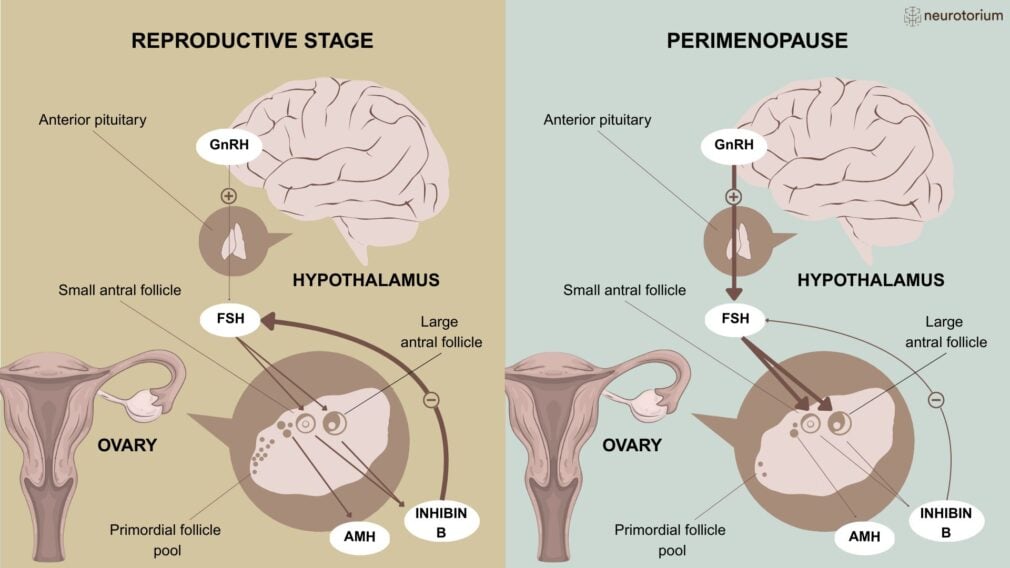
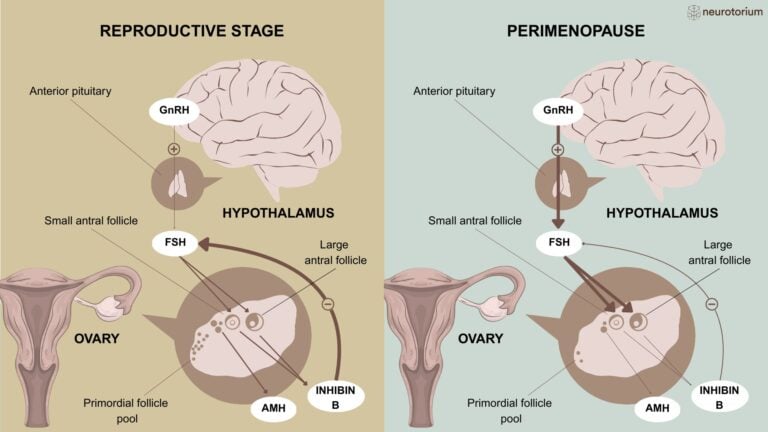
Figure 1: Physiological changes in the endocrine function during the perimenopause
A simplified illustration of the physiological changes in the endocrine function during the perimenopause. The follicle-stimulating hormone (FSH) is produced by the anterior pituitary in response to the gonadotropin-releasing hormone (GnRH) from the hypothalamus and released into the peripheral circulation.
Left Panel
During the reproductive years, FSH stimulates the maturation of ovarian follicles in the follicular phase of the menstrual cycle. The dominant follicle, in turn, secretes oestradiol and inhibin B, suppressing the secretion of FSH via negative feedback (thick lines on the left panel). The Anti-Müllerian hormone (AMH) produced by small growing ovarian follicles restrains the ovarian effects of FSH, and its plasma levels are correlated with the size of the ovarian reserve.
Right Panel
The ovarian reserve declines from about 250 thousand resting follicles at the time of puberty to less than 1000 by the final menstrual period. The steep decline in the ovarian reserve seen in the late reproductive stages/menopausal transition leads to a reduction of the concentrations of AMH and inhibin B, with loss of negative feedback on FSH (fine line on the right panel) and therefore increased release of FSH from the pituitary (bold arrows on right panel).
In the late menopausal transition, the hormonal compensatory mechanisms of previous stages fail. Extreme fluctuations in hormonal levels are associated with longer periods of amenorrhea (i.e., absence of menstrual periods), at least 60 days.1 Cycles are more likely to be anovulatory compared to previous stages and are underpinned by erratic hormonal patterns. On average, hyperestrogenism and decreased luteal phase progesterone are observed at this stage and may be associated with gynaecological symptoms. It has also been speculated that they may be linked to psychiatric morbidity.3,4 (Figure 2). Ovulatory cycles of normal length and hormonal profile, however, still occur in the late menopausal transition, suggesting there may be periods of fertility up to the final menstrual period.5 Compared to the duration of the early phase, that of the late menopausal transition is more consistent across people, ranging between one and three years approximately.
On average, hyperestrogenism and decreased luteal phase progesterone are observed at this stage and may be associated with gynaecological symptoms. It has also been speculated that they may be linked to psychiatric morbidity3,4
Only after 12 months without periods it is possible to establish that the final menstrual period, and therefore menopause, has occurred. The end of the late menopausal transition and the start of the early postmenopause stage are therefore determined retrospectively 12 months after the final menstrual period. The hormonal milieu of the early postmenopause is characterised by increasing levels of FSH and decreasing levels of oestrogen, that then stabilise approximately two years after the final menstrual period.
The term “perimenopause” covers the menopausal transition and the first year postmenopause, i.e., the first 12 months without periods.
Although evidence argues against a universal menopause syndrome6, vasomotor (hot flashes or night sweats) and urogenital symptoms are common. Vasomotor symptoms are usually framed as a gynaecological problem, but the underpinning mechanisms are thought to be centrally regulated by the hypothalamic kisspeptin-neurokinin B-dynorphin neuron complex.7
Menopause and mental health
Menopause has also been associated with a plethora of mental health problems, including depression, anxiety, psychosis, and suicidality. Methodological issues, however, often limit inference.8
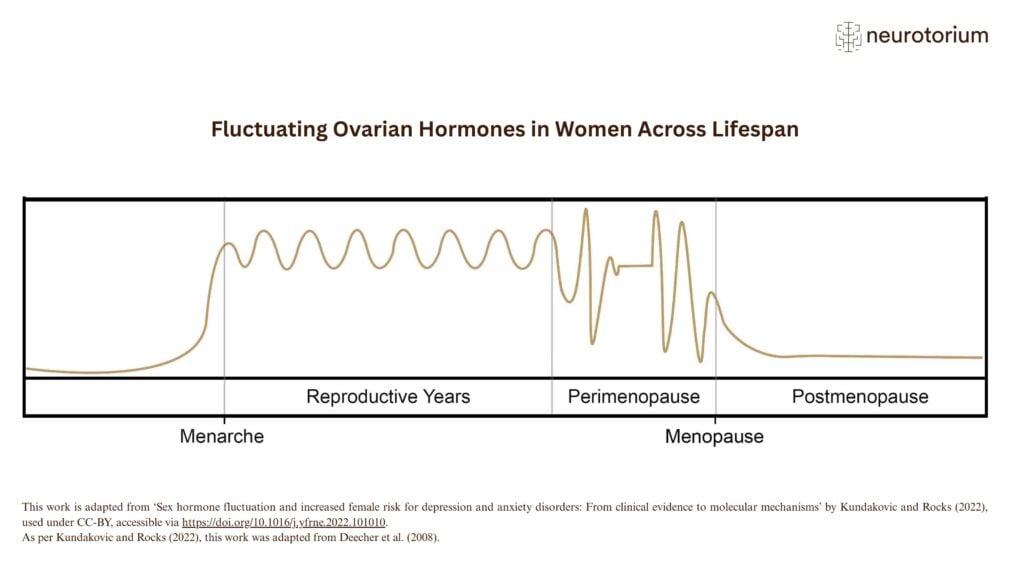
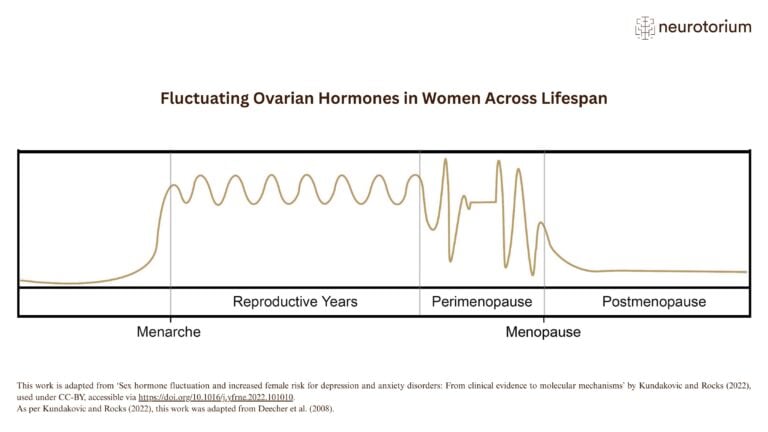
Figure 2: Fluctuating ovarian hormones in women across lifespan
A simplified illustration of ovarian sex steroid fluctuations across the female lifespan, from menarche (i.e., the first menstrual cycle) to menopause. For individuals with periods, menopause is typically marked by the final menstrual period, usually diagnosed after 12 months of amenorrhoea ( i.e., the absence of menstruation). Perimenopause is the transitional phase during which the first signs of ovarian function decline become apparent. It is a dynamic period characterised by changes in endocrine function, ovarian reserve, and menstrual cycle regularity.
Although STRAW defines perimenopause as a broader term than menopausal transition — extending to include the first year after the final menstrual period — the two terms are frequently used interchangeably in the literature.
Symptoms of depression, anxiety, and psychiatric disorders
Most studies on menopause and mental health have focused on the relative risk of high depressive symptom scores in the perimenopause compared to the premenopause, with mixed results.6 Some of the studies investigating depressive symptoms have also examined the occurrence of self-reported anxiety symptoms, again, with mixed results.6 Symptoms of depression and anxiety have often been assessed using self-administered questionnaires, with different cut-offs and follow-up periods, probably contributing to the heterogeneity of findings.
Prospective studies have identified subgroups of people who seem at higher risk of depressive symptoms during the menopausal transition (Table 1). The history of depression is the most robustly and consistently reported predictor of depressive symptoms during the menopausal transition.9-13 For women with history of depression, however, high depressive symptom scores are not limited to the perimenopause, but are reported across reproductive stages.6,9 Unsurprisingly, then, psycho-social factors that increase the risk of depression at perimenopause overlap with those of depression at other times.
Evidence also suggests a bidirectional association between vasomotor symptoms and both depression and anxiety.10,14,15 Vasomotor symptoms, however, can be hard to differentiate from the physical symptoms of anxiety. Moreover, they share risk factors with both anxiety and depression, making it difficult to disentangle causal relationships.16 For example, meta-analytic evidence suggests that sleeping problems account for the relationship between vasomotor symptoms and subsequent depression, but not vice-versa.17
Given the temporal relationship reported by some prospective studies, changes in ovarian hormones are obvious candidates to explain the possible association between perimenopause and depressive symptoms. Only a few mechanistic studies have, however, examined this hypothesis. Most evidence is indirect, based on animal models or observational studies, and suggests that sensitivity to fluctuations in ovarian hormones rather than hormonal concentrations per se trigger symptoms in people with a bio-psycho-social predisposition. Pre-clinical models, mostly based on male rats, have focused on neurosteroids, especially allopregnanolone, a metabolite of progesterone, which is believed to have a role in the GABAergic response to stress.16
In the absence of evidence-based mechanistic models, the prevention and treatment of bothersome depressive symptoms associated with perimenopause is based on expert opinions and a few randomized controlled trials. Approaches suggested by meta-analytic evidence of low-moderate grade include yoga18, mindfulness-based interventions19 and cognitive-behavioural20 therapies. Menopause hormone approaches are recommended by guidelines, but evidence is mixed.6,21 There is a variety of compounds, regimens, and administration routes, which should be tailored according to the person’s needs and response to treatment. For example, for people who have had a hysterectomy, oestrogen-only treatments may be prescribed instead of regimens including both oestrogen and progestogen. Moreover, according to guidelines, there are conditions, such as oestrogen-dependent cancers, that should preclude the use of hormone replacement therapy and others, for example migraine, that warrant caution.
In the absence of evidence-based mechanistic models, the prevention and treatment of bothersome depressive symptoms associated with perimenopause is based on expert opinions and a few randomized controlled trials
Depressive symptoms are clinically and probably aetiologically different from major depressive disorders.22 While previous smaller studies had not found any consistent evidence of an increased risk of the first onset of major depression during the perimenopause6, in a recent large, mostly cross-sectional, study including over 120 thousand UK Biobank participants, the incidence of first onset major depressive disorder was 30% higher (95% CI 16-45%) in the four years surrounding the final menstrual period than ten to six years before.23 In the prospective SWAN Mental Health sub-study, including 425 participants, trait anxiety and medical comorbidities were significant predictors of the first onset of major depression.24 The SWAN Mental Health sub-study also found that almost 60% of women with a prior history of major depressive disorder developed a new episode during the menopausal transition compared to less than 30% of women without.
There is a paucity of evidence on the incident risk of other psychiatric disorders. Together with major depression, the UK Biobank study also explored the association of mania and schizophrenia spectrum disorders with reproductive aging and found the largest effect size for mania (RR of 2.12, 95% CI 1.30–3.52).23 The incident rates of mania went back to premenopause levels in the postmenopause, suggesting a specific association between bipolar disorder and the perimenopause. Intriguingly, one of the most robust associations in psychiatry is that between another reproductive trigger, childbirth, and mania. For childbirth, however, the risk is an order of magnitude greater than for perimenopause.25 The UK Biobank study did not find any evidence of an association between the perimenopause and schizophrenia spectrum disorders. Only 19 out of 128,144 women, however, developed a schizophrenia spectrum disorder during the four years around the final menstrual period, suggesting that even larger samples may be needed to investigate non-affective psychotic disorders. Evidence on a link between perimenopause and other psychiatric disorders or suicidality is lacking, despite the claims of an association.6
Intriguingly, one of the most robust associations in psychiatry is that between another reproductive trigger, childbirth, and mania. For childbirth, however, the risk is an order of magnitude greater than for perimenopause25
Current Challenges in the Field and Potential Solutions
As only little is known about reproductive aging and mental health, there are a lot of opportunities for research to improve current knowledge. Research in this area is, however, challenging. Mechanisms are likely to be complex, involving bio-psycho-social factors and varying across groups of individuals. Such complexity would need to be reflected in the study design, requiring very large, well-characterised prospective samples. Differently from studies of the perinatal period, the duration of the perimenopause is unknown and may last from a few years to over a decade, making prospective assessment extremely costly and challenging. Moreover, preliminary evidence suggests that the effect sizes for perimenopause are smaller than those observed for the perinatal period.24 Passive data gathering from registry study or electronic health records does not provide the necessary information on reproductive aging. On the other side, studies specifically addressing reproductive aging often do not have the statistical power to detect rare, but devastating, outcomes such as psychosis or suicide and, in the long term, are prone to dropout. Future research will need to balance the tension between the necessity of very large samples and that of accurate prospective phenotyping.
As only little is known about reproductive aging and mental health, there are a lot of opportunities for research to improve current knowledge
| Menopause associated factors |
| Surgical menopause10,15 |
| Hysterectomy15,26 |
| Longer menopausal transition10,11 |
| Menstrual problems10 |
| Vasomotor symptoms12,14,16,27 |
| Medical factors |
| Higher body mass index11,13,14,28 |
| Poor health10,14,28 |
| Psycho-social factors |
| History of depression9-13 |
| Lower education14,28 |
| Lower socio-economic status / financial hardship11,12 |
| Poor social support12 |
| Stressful life events in the last 6 months12,13,27 |
| History of sexual abuse13 |
| Neuroticism29 |
| Family history of depression13 |
| Attitudes towards menopause/aging10,12 |
| Being single, divorced, widower28 |
| Poor sleep28 |
| Childhood adverse experiences30 |
Table 1: Risk factors of depressive symptoms during the menopausal transition according to prospective longitudinal studies. Only studies published after 2004 have been included as references.
Conclusion
To conclude, evidence suggests that there are subgroups of women at risk of developing mental health problems in the perimenopause/menopausal transition. More research is needed to identify mechanisms, risk factors, prevention, and treatment strategies. Clinical management should consider that mechanisms are likely to be complex and heterogeneous. Therefore, individualised multidisciplinary approaches may be necessary.
Clinical management should consider that mechanisms are likely to be complex and heterogeneous