The exposome and health
Health is increasingly understood as the cumulative product of lifelong exposures. This “exposome” complements the genome by encompassing all environmental influences from conception onwards1-3. First conceived in environmental health research, it now includes both physical exposures (e.g. air pollution, toxins, infections, diet, climate) and social exposures (originally understood as social determinants of health; e.g. family environment, education, socioeconomic status, discrimination, neighborhood factors) across the life course1,4. The exposome is more than a collection of isolated risk factors. It brings together multiple exposures that accumulate and interact to shape individual trajectories. This holistic lens is especially relevant for complex conditions like dementia, where compound exposures drive vulnerability.
First conceived in environmental health research, it now includes both physical exposures (e.g., air pollution, toxins, infections, diet, climate) and social exposures (originally understood as social determinants of health; e.g., family environment, education, socioeconomic status, discrimination, neighborhood factors) across the life course1,4.
Many studies have identified modifiable factors such as education, hypertension, social interaction, smoking, and air quality, usually considered separately. By contrast, the exposome considers their totality and interplay. The social exposome highlights how living conditions, cultural context, and policies influence brain aging alongside traditional biomedical factors1,4,5. This perspective aligns with the recognition that social determinants are as fundamental to aging as molecular and genetic factors. Adversity6, low socioeconomic status7, and chronic stress8 are recognized as social influences on aging, analogous to biological influences9. In sum, the exposome encourages clinicians and researchers to see brain health as the culmination of lifelong physical and social influences, not just genes or late-life events.
The social exposome highlights how living conditions, cultural context, and policies influence brain aging alongside traditional biomedical factors1,4,5.
Physical and social exposome in aging and dementia
Dementia risk is influenced by exposures across the entire lifespan, from prenatal life to older age. Early-life conditions set the stage for later health. Adverse childhood experiences (abuse, neglect, poverty, malnutrition, and household dysfunction) are linked to poorer late-life cognition and higher dementia risk in a dose-dependent manner5,10. Lower education (often rooted in early socioeconomic disadvantage) is linked to higher dementia risk, likely through reduced cognitive reserve5 as well as brain reserve11,12. Reserve reflects pre-existing brain or cognitive resources that buffer pathology, whereas resilience is the dynamic capacity to adapt and maintain function amid aging, stress, or disease13. Individuals with multiple childhood adversities have significantly higher odds of dementia, even after adjusting for other factors10. Chronic stress and trauma accelerate cellular aging, with epigenetic clock acceleration and telomere shortening as biomarkers of ageing14. Adults early exposed to trauma often show older epigenetic profiles14. Early stress primes inflammatory and neuroendocrine pathways, with elevated cortisol and immune dysregulation damaging the brain15. Those with childhood abuse or neglect show long-term increases in inflammatory markers, altered HPA activity, and structural brain differences15,16.
Mid-life and late-life exposures are different but also important. Some of the risk factors highlighted by the Lancet Commission17 (e.g., social isolation, air pollution) lie within the broader exposome1,4,5. Structural socioeconomic inequality (i.e., as measured by the Gini coefficient, a measure that quantifies the unequal distribution of income or wealth within a population) is linearly associated with the brain structure and function in aging and dementia18,19. Individual physical aspects matter too. Nutritional deficiencies, infections, and toxic exposures (lead, air pollutants, pesticides, plastics) over decades contribute to direct and indirect cerebrovascular damage and neurodegeneration20. Long-term exposure to fine particulate air pollution predicts faster decline21 and accelerated brain age in older adults and people with dementia22. Moreover, the combination of physical and social exposomes induces much more accelerated aging than individual factors. An international study across 40 countries showed that people living with higher pollution, inequality, and disease burden had much more accelerated brain aging4. Similar findings have been observed using brain age clocks derived from neuroimaging in aging and dementia22. Accelerated aging (also called aging clocks22,23) refers to the biological estimation of an organ’s (or system’s) age and its deviation from chronological age, typically derived from computational models applied to large datasets. This concept should not be confused with the outdated notion that dementia represents a form of normal aging.
Structural socioeconomic inequality (i.e., as measured by the Gini coefficient, a measure that quantifies the unequal distribution of income or wealth within a population) is linearly associated with the brain structure and function in aging and dementia18,19.
Across all stages, cumulative and synergistic burdens drive brain aging5. Importantly, not all exposed individuals develop dementia. Resilience factors (i.e., social, genetic, or exposome factors that are protective against the effects of insult, injury, or exposure) buffer against risk. Such protective factors include lifelong cognitive engagement, strong social support, physical activity, and quality healthcare13. Potential mechanisms through which these factors confer protection include epigenetic reprogramming, enhanced neuroplasticity, neuroendocrine modulation, immune–inflammatory signaling, metabolic and vascular regulation, microbiome alterations, and allostatic interoceptive processes13. However, it remains unclear whether these factors slow the biological disease processes underlying dementia or instead confer resilience against their effects13. Mentally stimulating activities in mid-life may mitigate educational deprivation’s effects24. Protective factors such as multilingualism and healthy habits also mitigate accelerated aging25,26. Structural factors such as healthcare infrastructure and social policies also modify outcomes, leading to heterogeneity in risk trajectories. Ultimately, dementia emerges from the interplay between adverse and protective exposures, just as for adverse and protective genomes, making the exposome a driver of risk and a potential target for prevention and intervention across the lifespan.
However, it remains unclear whether these factors slow the biological disease processes underlying dementia or instead confer resilience against their effects13.
Current progress and challenges
Recognition of the exposome’s role in dementia has spurred advances (Figure 1). Epidemiological studies increasingly integrate multi-exposure data. Longitudinal cohorts like the Health and Retirement Study and UK Biobank link cognition with life-course data enables exposome-wide analyses27. Composite indices such as the Social Vulnerability Index, exposome-wide association studies (ExWAS), and polysocial risk scores synthesize multiple socioeconomic and environmental variables. These indices consistently associate with worse cognition and dementia incidence, validating disadvantage as a multidimensional factor. Social exposome analyses have identified hundreds of factors influencing mental health, cognition, function, and reserve5. This multifactor approach marks progress beyond single-exposure studies. At the biological level, growing evidence shows that the concept of biological embedding of the exposome is substantiated by associations between adverse exposures and a range of biological markers. Chronic inflammation, stress hormones, epigenetic acceleration14, high allostatic load8, biobehavioral clocks4, and neuroimaging markers of accelerated aging22 provide evidence of pathways linking exposures to dementia. Multi-omics technologies (metabolomics, proteomics, transcriptomics) are increasingly used to identify molecular signatures of the exposome28.
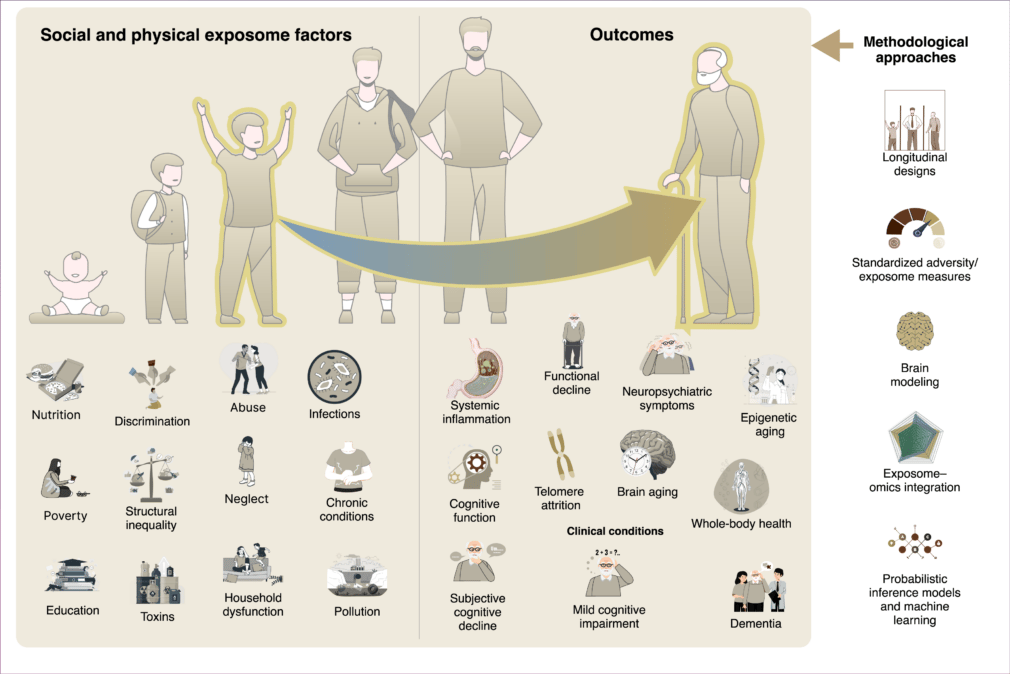
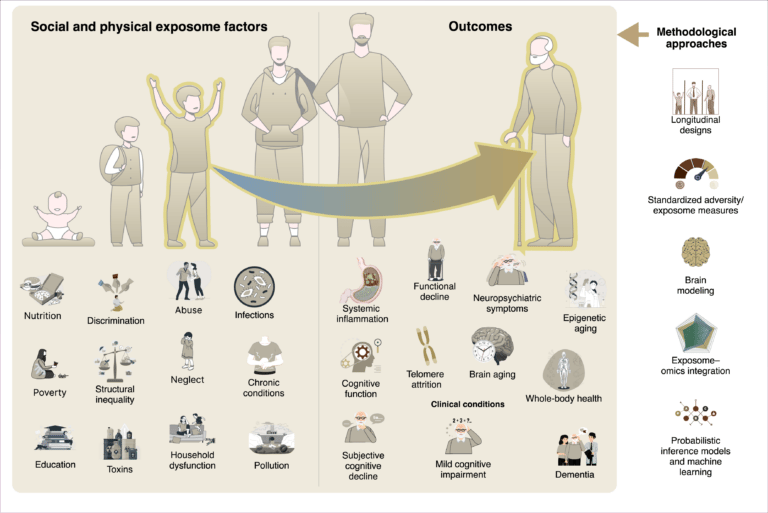
Figure 1. The physical and social exposome in brain health and dementia across the lifespan
Social and physical exposome factors (e.g., nutrition, poverty, education, discrimination, abuse, infections, toxins, pollution, structural inequality) accumulate across development and adulthood, shaping biological and clinical outcomes. These exposures become biologically embedded through pathways including systemic inflammation, telomere attrition, cognitive decline, accelerated aging, and whole-body health impairments. Such processes contribute to functional decline, neuropsychiatric symptoms, subjective cognitive decline, mild cognitive impairment, and ultimately dementia. Methodological approaches—such as longitudinal designs, standardized exposome/adversity measures, brain modeling, exposome–omics integration, and probabilistic inference models are required to capture the complexity of these dynamics.
However, multiple challenges remain. The exposome is vast and aspects of it can be difficult to quantify, spanning domains and time points. The heterogeneity of measures (income, education, composite indices), hinders comparisons. Standardized metrics for social and environmental exposures are needed9. Efforts to harmonize measures, such as standardized questionnaires for childhood adversity or social isolation, are underway but remain limited5. Capturing life-course dynamics is another challenge. Many cohorts start recruitment in mid- or late-life, missing early exposures or relying on retrospective reports prone to bias10. Long-term cohorts that follow individuals from youth to old age (e.g., the 1946 birth cohort or the Lothian Birth cohort in the UK) are rare but invaluable. Creative data linkages (i.e., childhood school records with dementia registries) could fill some these gaps.
Diversity is another major gap in current data and models. Most evidence on the impact of the exposome comes from high-income countries, even when including disadvantaged socioeconomic groups within the high-income countries. There is limited data on exposomal influences in long-term cohorts from Africa, Latin America, South Asia, and elsewhere. These regions face different exposures such as infectious disease, displacement, and violence. Such exposomes may shape dementia risk more severely, or differently. Without inclusion of diverse cohorts with differing exposomes, the generalizability of outcomes and equity suffer. Ensuring global representation is both scientific and ethical.
The conceptual distinction between the exposome and the genome is heuristic rather than absolute. The exposome interacts with the individual genetic and biological background. For instance, genetic variants influencing addiction, impulsivity, or attentional control may modulate risk-taking behaviors and educational attainment, thereby altering one’s adaptation to exposome. Conversely, environmental and social exposures may modify gene expression through epigenetic mechanisms, reinforcing the bidirectional interplay between genome and exposome. These is particularly relevant for dementia, requiring integrative models that capture dynamic genome–exposome interactions29.
For instance, genetic variants influencing addiction, impulsivity, or attentional control may modulate risk-taking behaviors and educational attainment, thereby altering one’s adaptation to exposome.
Causal inference is difficult to assess when studying the exposome. Poverty often co-occurs with poor nutrition and stress, complicating causal attribution. Techniques such as Mendelian randomization and quasi-experimental designs can help but require further adaptation to social and environmental factors. Finally, translation into interventions and policy remains limited. Most of the social exposome factors (poverty, poor education, violence, social isolation) lie outside healthcare’s scope and require broader social policy solutions. Collaboration across disciplines and intersectoral planning is essential. At the same time, care is needed to avoid stigmatizing individuals by labeling them high-risk without offering support.
Most of the social exposome factors (poverty, poor education, violence, social isolation) lie outside healthcare’s scope and require broader social policy solutions. Collaboration across disciplines and intersectoral planning is essential.
The future
Future research will benefit from being integrative and global. Comprehensive exposomic datasets linking social, environmental, biological, and clinical data across longitudinal assessments are needed. Combining epidemiological cohorts with environmental data (satellite pollution, geocoded neighborhoods) and multi-omics will help disentangle causation and refine hypotheses for the interventional studies and risk-reduction strategies. Advanced computational pipelines (e.g., generative biophysical models of brain dynamics informed by exposome parameters) and systems science approaches (e.g., directed acyclic graphs or Bayesian networks) can detect nonlinear interactions and multidimensional patterns beyond the capabilities of traditional statistical models5,30. Exposome-genome interactions29 are much needed to understand truly synergetic effects. Such tools may enable personalized risk prediction and tailored interventions.
Interdisciplinary collaboration from environmental science, sociology, neurobiology, and data science will be essential. Growth in exposome-omics studies, analyzing social and environmental data alongside molecular signatures, may reveal mechanistic pathways and therapeutic targets. Standardization will also shape the field. Core exposome measures, such as validated instruments for social determinants and physical exposures, should be integrated into dementia research globally. Organizations like WHO, the FDA, and NIA already promote harmonization9. More diverse, global cohorts will emerge, including registries in low- and middle-income countries and pooled cross-cohort collaborations.
Research must move from observational associations to evidence-based prevention strategies. The promise of exposome science lies in reducing harmful exposures and bolstering protective ones. Future trials may test multi-domain interventions targeting both social and physical factors. Policy-level actions informed by exposome findings, such as investing in education, enforcing clean air, and strengthening social safety nets will be crucial for long-term brain health. Identifying high-risk profiles should mobilize collective resources, not seek to blame individuals at risk. Engaging communities with high trustworthiness, building capacity in underrepresented regions, and recognizing resilience factors13 such as social cohesion will be essential for success.
Advanced computational pipelines (e.g., generative biophysical models of brain dynamics informed by exposome parameters) and systems science approaches (e.g., directed acyclic graphs or Bayesian networks) can detect nonlinear interactions and multidimensional patterns beyond the capabilities of traditional statistical models5,30.
|
|
|
|
Table 1: Key Points
Conclusions
The exposome has become central to understanding brain health and dementia (Table 1). It compels us to move beyond single risk factors and instead consider how cumulative exposures shape the aging brain. Childhood adversity, mid-life risks, and late-life social stressors all leave biological imprints that accelerate brain aging and dementia. These insights underscore unhealthy aging and dementia as not only neurological but also social, environmental, and structural phenomena. Yet evidence remains fragmented, with methodological and equity challenges. To realize an exposome-informed framework, the field must pursue harmonized measures, longitudinal and diverse studies, and integrative analyses. Most importantly, new avenues for prevention should intervene at multiple levels, addressing upstream determinants and mitigating downstream consequences. Embracing the exposome in research and practice ultimately aims to promote equitable brain health worldwide.
These insights underscore unhealthy aging and dementia as not only neurological but also social, environmental, and structural phenomena. Yet evidence remains fragmented, with methodological and equity challenges.