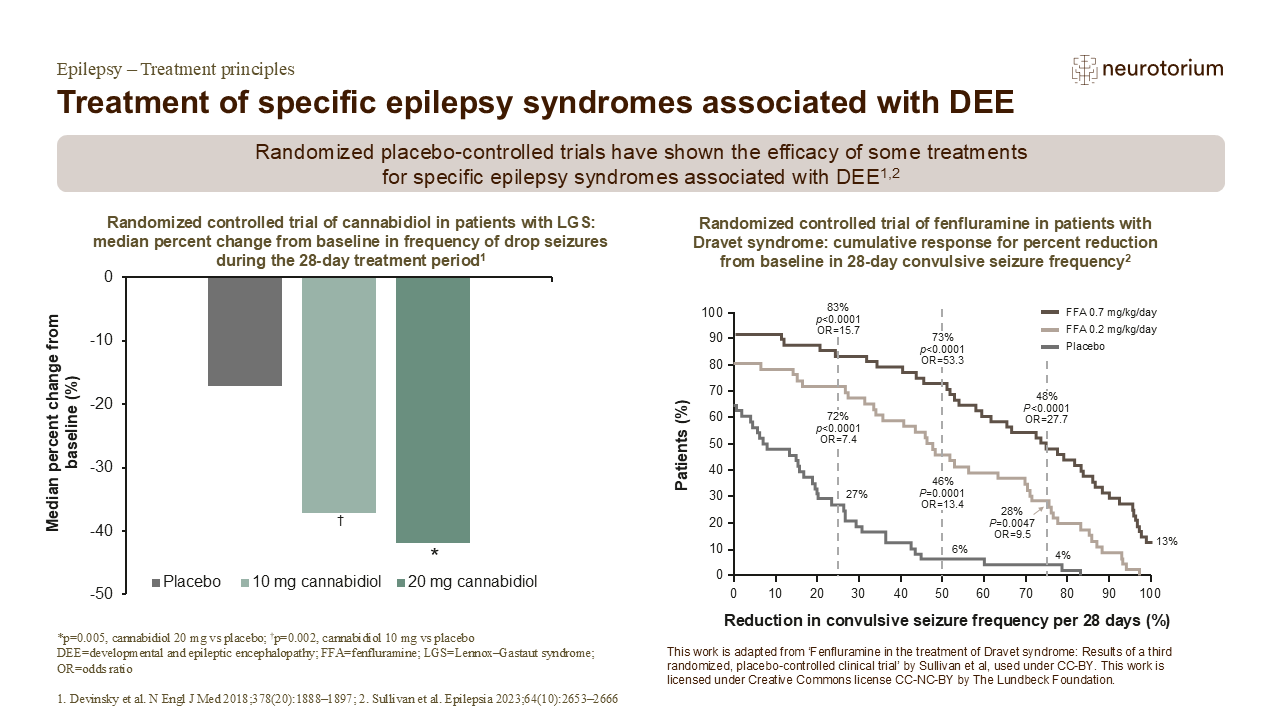
The efficacy of cannabidiol in 225 children and adults with LGS was assessed in a randomized controlled trial.1 At baseline in the 76 patients assigned to the 20 mg cannabidiol group, 47% of participants were taking clobazam, 37% were taking valproate, 32% were taking levetiracetam, 26% were taking lamotrigine, and 34% were taking rufinamide.1 The median number of drop seizures during the 28-day baseline period in the overall population was between 80 and 87.1
The percentage of participants who experienced at least a 50% median reduction from baseline in the frequency of drop seizures during the 28-day treatment period was 39% in the cannabidiol 20 mg group, 36% in the cannabidiol 10 mg group, and 14% in the placebo group.1
In a separate trial, the efficacy of two doses of adjunctive fenfluramine were evaluated in 143 children and adolescents with Dravet syndrome whose seizures were not adequately controlled by their current ASM regimen.2 At baseline, 56% of participants were taking clobazam, 28% were taking levetiracetam, 27% were taking topiramate, and 58% were taking valproate.2 Compared with placebo, the cumulative frequency of convulsive seizures per 28-day period decreased consistently and significantly over time with both doses of fenfluramine.2
References: