In terms of altering the current environment towards positive and meaningful change for patients diagnosed with brain diseases such as Alzheimer’s disease, multiple sclerosis, Parkinson’s disease, motor neurone disease, and frontotemporal dementia, we must identify previously unrecognized bottlenecks that continue to negatively impact the translational process. Inherent disease characteristics, such as clinical heterogeneity and different mechanistic processes, make a one-size-fits-all solution unworkable (Fig. 1). As such, clinical trial durations remain unnecessary long, most often due to a lack of proven biomarkers and the choice of appropriate trial outcome measures. Progress in the current era of potential therapeutic advance, in which an unparalleled number of disease-modifying therapies are ready to be trialled or implemented into clinical practice, requires the establishment of stronger collaborative networks across clinical and translational neuroscience domains.
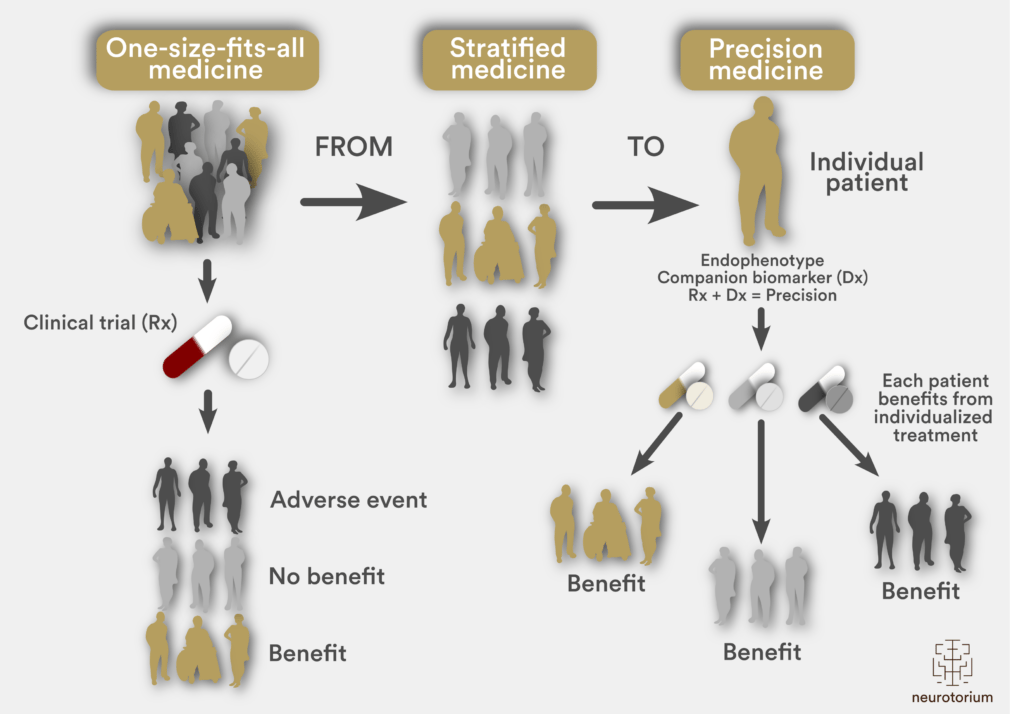
Figure 1: From One-Size-Fits-All to Precision Medicine
The use of a single treatment in heterogeneous populations has proved problematic in clinical trials undertaken in patients with neurodegenerative diseases. This one-size-fits-all approach (left) leads to a scenario whereby some patients may benefit from therapy but others do not. Stratified medicine (centre) improves on the approach by enabling patients to be separated into more homogeneous groups based on demographics, clinical phenotype and molecular subtypes. More recent advances in technology are enabling a transition to a precision medicine paradigm (right), incorporating phenotypic and genotypic data to guide individualized therapy.
So, let’s consider some of the important next steps necessary to translate recent molecular discoveries into the development of better treatments and thereby outcomes, for patients diagnosed with neurodegenerative disease.
Step 1: A precise approach
In light of the heterogeneity of clinical presentations, allied with the underlying molecular complexity that determines the varied expression of neurodegenerative disease, the concept of therapy that is tailored to an individual’s genetic signature has started to emerge. In consort, the understanding of individual differences in response to a trial therapy, particularly in a longitudinal manner to assess progression, requires reliable biomarkers. Several candidate biomarkers for brain disease have emerged, including biofluid, neurophysiological, and neuroimaging measures that indicate various aspects of disease, from network-level dysfunction to structural changes and basic cellular processes.6-9 However, their clinical utility and ability to enable patient stratification remain variable. While structural changes involving the brain can be observed with magnetic resonance imaging (MRI), the sensitivity of imaging measures to longitudinal change is inconsistent even at the group level, while functional changes are even more difficult to quantify. Emerging techniques that combine neuroimaging with functional neurophysiological approaches, such as transcranial magnetic stimulation and electroencephalography may yet provide utility to capture systems-level signatures for neurodegenerative diseases at a network level.
Across the neurodegenerative diseases, measurement of neurofilament in the cerebrospinal fluid (CSF) and blood represent promising biofluid markers of disease.10 Currently, it seems likely that neurofilament levels correlate with the rate of disease progression and are most strongly associated with the level of brain involvement, as compared to spinal cord and peripheral nerve manifestations. CSF and serum levels of the neurofilament light chain (NfL) correlate tightly with one another and serum assays have increased in sensitivity to offer a simple, non-invasive measure. As an exemplar, measurement of serum NfL has been applied in trials of the latest therapies for multiple sclerosis. In the context of monitoring treatment responses in trials of therapies for neurodegeneration, failure of serum NfL levels to decrease during treatment may enable decisions regarding the efficacy of potential therapeutic agents to be made earlier.
Across the neurodegenerative diseases, measurement of neurofilament in the cerebrospinal fluid (CSF) and blood represent promising biofluid markers of disease.10
In a multi-arm, multi-stage (MAMS) platform trial structure, patients could subsequently switch to a new trial agent. Separate to acute treatment approaches, biomarkers will also be needed to enable the testing of preventive strategies for at-risk individuals for neurodegenerative diseases, including dementia. Currently, the identification of individuals who are at risk of developing dementia can only be done by screening individuals from families that carry highly penetrant mutations. However, some data suggest that predictive biomarkers do exist. As an example, a large primary care cohort study identified novel metabolic markers, including alterations in carbohydrate, lipid and apolipoprotein profiles, that were associated neurodegenerative disease.
Step 2: Genetic and molecular therapy
The success of targeted therapies for the predominantly paediatric neurodegenerative disease spinal muscular atrophy, antisense oligonucleotides (ASOs) or small molecules, and the use of viral vectors for gene replacement, has demonstrated the power of genetic therapies.11 Genetic mutations are present in varying degrees across the spectrum of the more common neurodegenerative diseases, although the multiplicity of mutation types that cause disease, including missense mutations, repeat expansions, and loss-of-function mutations, means that a variety of gene therapy approaches will likely be needed to target all causal mutations.
In addition, although many causal mutations are associated with the toxicity of the mutant protein, and therefore reducing the expression of the toxic protein seems a logical approach, many different mutations have been identified in single genes, making it difficult to design ASOs to selectively target the mutant allele.
Separate to the scientific basis of genetic therapy for neurodegenerative disease, there remains further consideration regarding the optimal means of therapeutic delivery (Fig. 2). To maximize the benefits of these ASO therapies, various routes of administration have been considered, from systemic intravenous administration to intraparenchymal brain delivery, although intrathecal delivery has been used most frequently to date.
Separate to the scientific basis of genetic therapy for neurodegenerative disease, there remains further consideration regarding the optimal means of therapeutic delivery.
Commonly observed lumbar puncture-related adverse events could be mitigated in the future through the development of specialized nanotechnology drug delivery systems. The efficiency of ASOs is currently limited by a failure to traverse the blood–brain barrier, a generic problem that will need to be solved for all forms of precision-based molecular therapies. However, ASOs are also limited by complexities in intracellular processing that limit their direct engagement with mRNA species. Advances in the development of chemical modifications that increase the stability of ASOs and their ability to penetrate cells will inevitably promote a new generation of ASOs that could be delivered via the systemic circulation.

Figure 2: Drug Delivery Systems to the Brain
Brain drug delivery systems, allied with their characteristics for overcoming the blood-brain barrier (BBB). Implantable delivery ensures that drug preparations are directly implanted into the brain by surgery or through special instruments. In cell penetrating peptide mediated BBB crossing, positively charged particles are modified onto the surface of the preparations to heighten the interactions. Temporary disruption of BBB can currently be achieved through ultrasound or magnetic disruption technologies. Intranasal delivery enables drug preparations to cross the olfactory mucosa in the nasal cavity, to be transported into the brain. Finally, molecules, ligands or substrates can be modified onto the surface of the preparations via receptors, carriers or transporter-mediated approaches.
Expectations in most therapeutic trials in neurodegeneration are limited to slowing or arresting disease progression, but patients understandably place a high value on the search for treatments that could restore function.12 Whether targeting the cause of disease will influence disease progression in symptomatic individuals remains unclear; if this is not the case, then gene therapy that targets causal genes would be pointless in these people. So-called regenerative medicine approaches, in which various forms of stem cells or neural progenitor cells are used, are based around the notion that transplanted cells could provide trophic support to slow the disease process and raise hopes for restorative therapy for people already living with neurodegenerative diseases. However, given the immense complexity of the functional networks required to produce clinical improvements and the fact that these networks are produced via a complex developmental programme that is largely inactivated in adulthood, the restoration of function with cellular therapy is currently a theoretical rather than practical option.
Step 3: Improving trial design
Across neurodegenerative diseases, the mainstay of clinical research has been the randomized controlled trial, in which the effects of a potential therapeutic candidate are compared with those of a placebo. Typically, a single agent is tested in a homogeneous population, with a null hypothesis being that treatment with this agent is no better than standard care. Such trials are costly and inefficient given that only a small proportion of treatments make it from initial testing to the commercial market. As an example, the FDA reports that just 8% of treatments tested in a phase I trial make it to market.13
Typically, a single agent is tested in a homogeneous population, with a null hypothesis being that treatment with this agent is no better than standard care.
The inefficiencies in the current approach to clinical trials become more evident when considering the time commitment and expense required for staff recruitment, deployment and training, contract negotiations, regulatory approvals, monitoring arrangements, and infrastructure; these costs must be reconstituted anew for each trial. After trial completion, staff move elsewhere, skills are lost, and approvals lapse. A further inefficiency, and perhaps the most problematic from a patient perspective, is the fact that each study has a separate placebo arm. Such an approach is costly and is a major disincentive for patients to participate in a clinical trial, particularly as their disease is terminal.
Interest is increasing in methods that maintain the rigour of randomized controlled trials while maximizing their efficiency, reducing costs, and providing answers regarding efficacy. One approach that is gaining traction is the use of a master protocol for the simultaneous evaluation of multiple compounds.14-15 With a master protocol, a common system is used for patient selection, logistics, templates, and data management. This approach can incorporate precision genotyping and molecular markers and can be implemented in different trial designs, including basket trials (in which the therapeutic target is a specific genetic mutation) and MAMS platform trials. Such designs have been successfully used in cancer, HIV and some neurological diseases such as multiple sclerosis.16-17
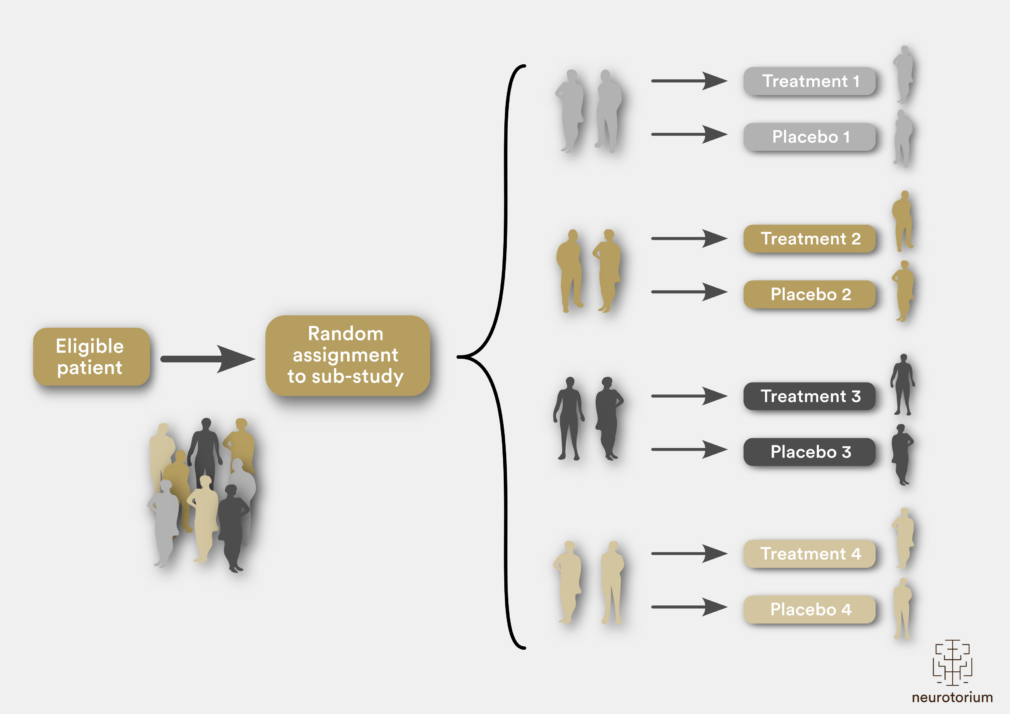
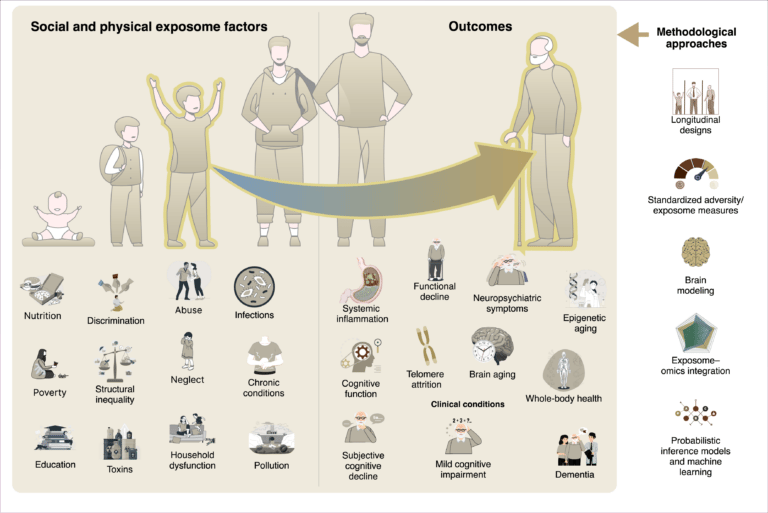
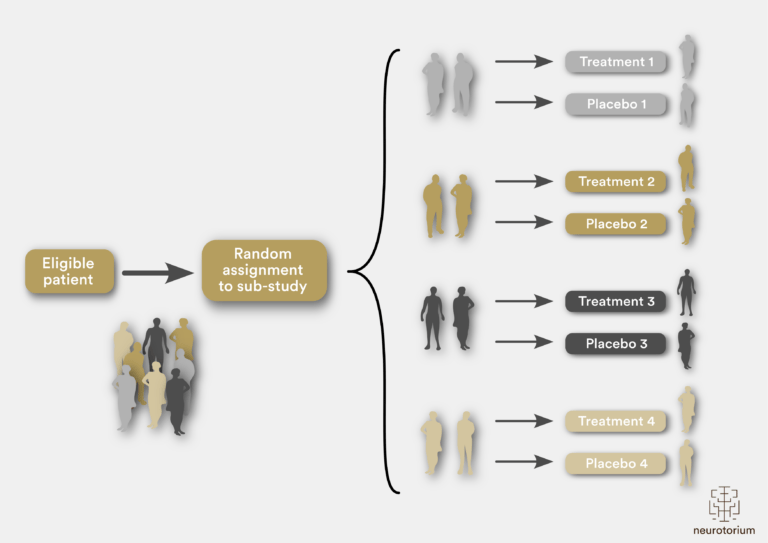
Figure 3: A multi-arm, multi-stage platform trial illustration
Protocol for a multi-arm, multi-stage platform trial, whereby patients are randomly assigned to one of four sub-studies and then further randomly assigned to receive active treatment or placebo. Such a protocol determines patient selection and outcome measures, incorporating biomarkers across the different sub-studies. Pre-planned interim analyses can be incorporated into the trial design, such that if a specific treatment is established to have a favourable efficacy signal, this arm can then seamlessly move into phase III. Alternatively, if futility criteria are met, specific treatment arms can be dropped, with new arms then added over time.
In the context of disease heterogeneity, the stratification of patients as an inclusion criterion for clinical trials is becoming an important consideration when determining the efficacy of a compound. A lead-in phase during which patients are monitored before being randomly assigned to receive a treatment (typically after patient inclusion but before initiation of therapy) might provide a more accurate indication of disease progression in individuals and thereby enable the assessment of investigational drug efficacy.18-20 In addition, patient stratification can be based on clinical parameters, genetics, disease stage, and disease trajectory.
The purpose of pre-selection criteria for trial entry is to facilitate the use of tailored treatments. Some studies suggest that modifier genes, such as those that affect prognosis, can also be used as enrolment criteria, and specific clinical features, such as respiratory, nutritional, or emotional symptoms, have been used for pre-selection. Stratification at the time of recruitment also enables patients to be matched according to their likely clinical course. However, across the various disease states, the clinical course remains heterogeneous and unpredictable and, therefore, attempts to homogenize cohorts on the basis of likely disease course at trial entry lead to restrictive inclusion criteria for most trials. Another emerging approach to stratification for clinical trials is disease staging. Staging is important because the various disease states represent a continuously evolving process, and treatments might need to be administered at precise time points in the disease course to be beneficial. Disease staging provides information about this timing.
Another emerging approach to stratification for clinical trials is disease staging.
Separately, the requirement for participants in clinical trials to make episodic visits to study centres so that progression of their disease can be measured imposes substantial restrictions on the conduct of clinical trials. First, it limits participation to patients who live close enough to a study centre to attend mandatory visits on a monthly or bimonthly schedule. In an analysis of the reasons for poor adherence in clinical trials, travel difficulties and caregiver burden were contributing factors to the withdrawal of 27% of patients who withdrew their consent.21 Second, the requirement for visits to study centres means that substantial variability is seen in outcome measures that are commonly used in phase II and III trials; some of this variability might be due to true fluctuations in patient status and some due to measurement error. Both of these issues could be addressed if data could be collected through more frequent assessment of patients because it would reduce variability by increasing the sampling frequency; however, this would be possible only if undertaken in patients’ homes.
Other aspects of trial participation, such as safety assessments and drug dispensing, could also be accommodated at patients’ homes, thereby enabling the participation of patients who have previously been deemed inaccessible. The value of self-reported outcomes has previously been considered, whereby patients directly entered their symptoms and the data were subsequently analysed.22 Such patient-reported outcomes are valuable as they are cost-effective, can be implemented from a distance, can provide comprehensive assessments, and can capture an element of personal well-being that is not captured with other approaches. The limitations of self-reported outcomes include a lack of objectivity and unfamiliarity with the use of digital platforms that are necessary for this approach to be successfully implemented. Home-based outcome measures are also becoming increasingly relevant for patients with neurodegenerative diseases who have substantial disease-related disability, particularly in the context of a shift towards telemedicine owing to the COVID-19 pandemic.
Other aspects of trial participation, such as safety assessments and drug dispensing, could also be accommodated at patients’ homes, thereby enabling the participation of patients who have previously been deemed inaccessible.
This video delves into the complexities of clinical trials and treatment for dementia. Professor Dr. Matthew Kiernan discusses the limitations of current trial models, the need for personalized biomarkers, and the challenges of using a one-size-fits-all approach.
Conclusion
The advances in clinical trial models seem critical to accelerate the translation of novel therapies into the clinic.5,23 The implementation of new and evolving approaches for the assessment of current and future treatment strategies across neurodegenerative diseases and dementia could significantly shorten trial durations and reduce costs and burden on patients, thereby providing hope that effective therapies can be rapidly translated into the clinic.24-26